Team:TU-Delft/Project/Hydroxylation
From 2011.igem.org








-
 Home
Home
-
 The Project
What we are doing
The Project
What we are doing
-
 The Team
Who we are
The Team
Who we are
-
 Notebooks
What we did
Notebooks
What we did
-
 Human Practice
Awareness
Human Practice
Awareness
-
 Safety
Responsibilty
Safety
Responsibilty
Hydroxylation
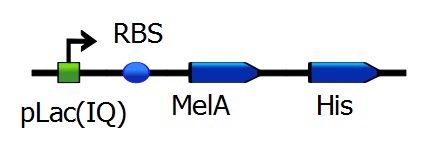
In order to make the Mfp-5 on the outer membrane of the E. coli cells sticky, an hydroxyl group has to be added to the tyrosinases present in Mfp-5. For this we use the enzyme melA, which conversts tyrosine to L-DOPA. For this, we use Biobricks made by the iGEM team of Tokyo Tech 2009 (BBa_K193601 and BBa_K193602). To be able to analyse the tyrosinase activity of MelA, we needed to have purified MelA. For this, we decided to add a His-tag to the MelA protein by the use of PCR. The PCR reaction was followed by digestion and ligation to put the sequence of MelA-His back into a BioBrick backbone. Since we strive towards a final system in which no additional compounds are needed for adhesiveness., tyrosinase production should also be done by the Mfp-5 producing cells. An initial approach would be introducing both genes, as well Mfp-5 production as MelA production, in one or two separate plasmids. This will probably cause inclusion bodies due to internal activation of the adhesiveness. To prevent this, tyrosinase may be transported to the periplasm using the same Sec-transporter as described earlier. Although it would likely be degraded rapidly by the largely present proteases, activation may still occur. Proper integration of this mechanism is definitely a promising achievement for future research.
Retrieved from "http://2011.igem.org/Team:TU-Delft/Project/Hydroxylation"
 "
"