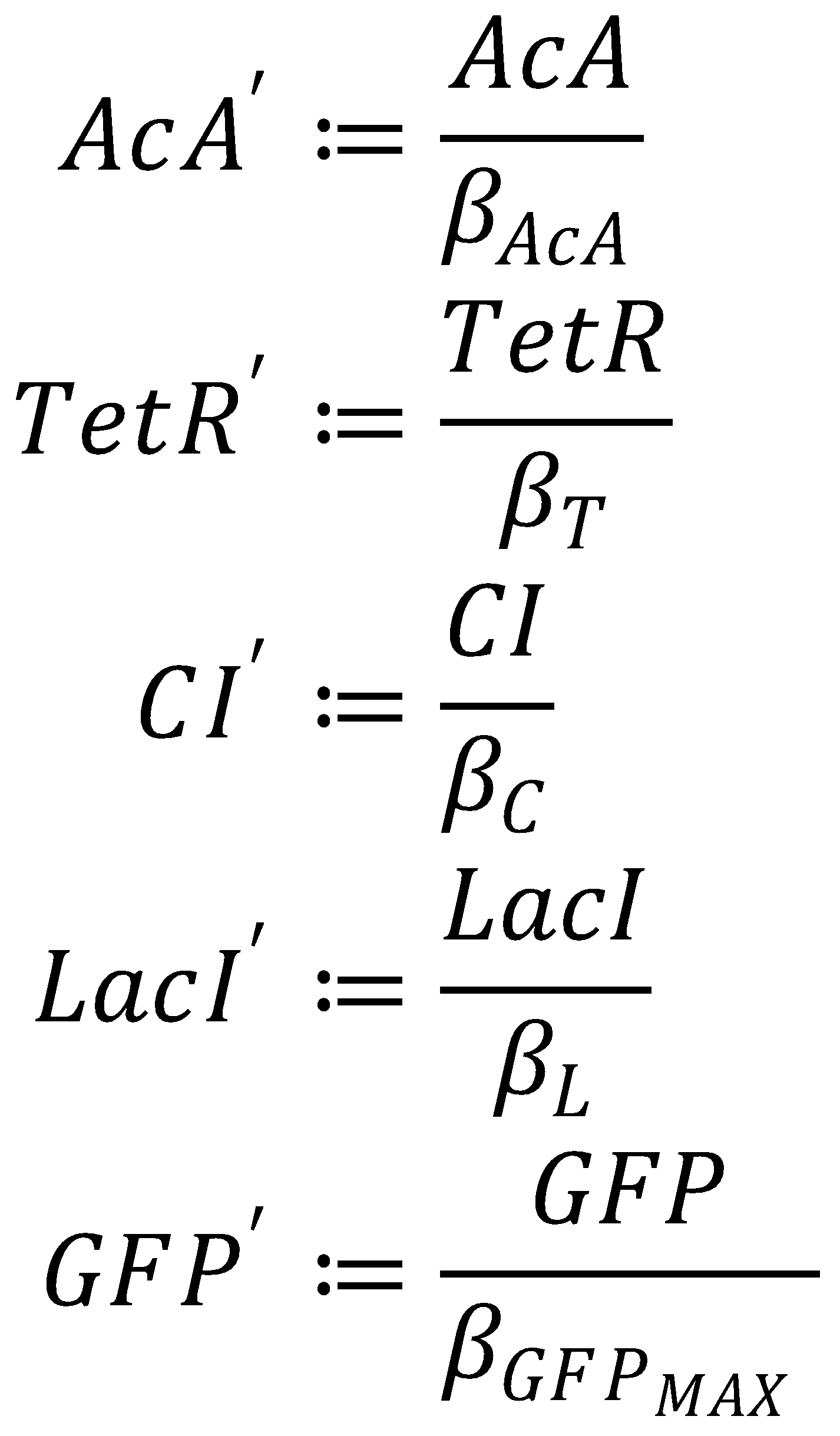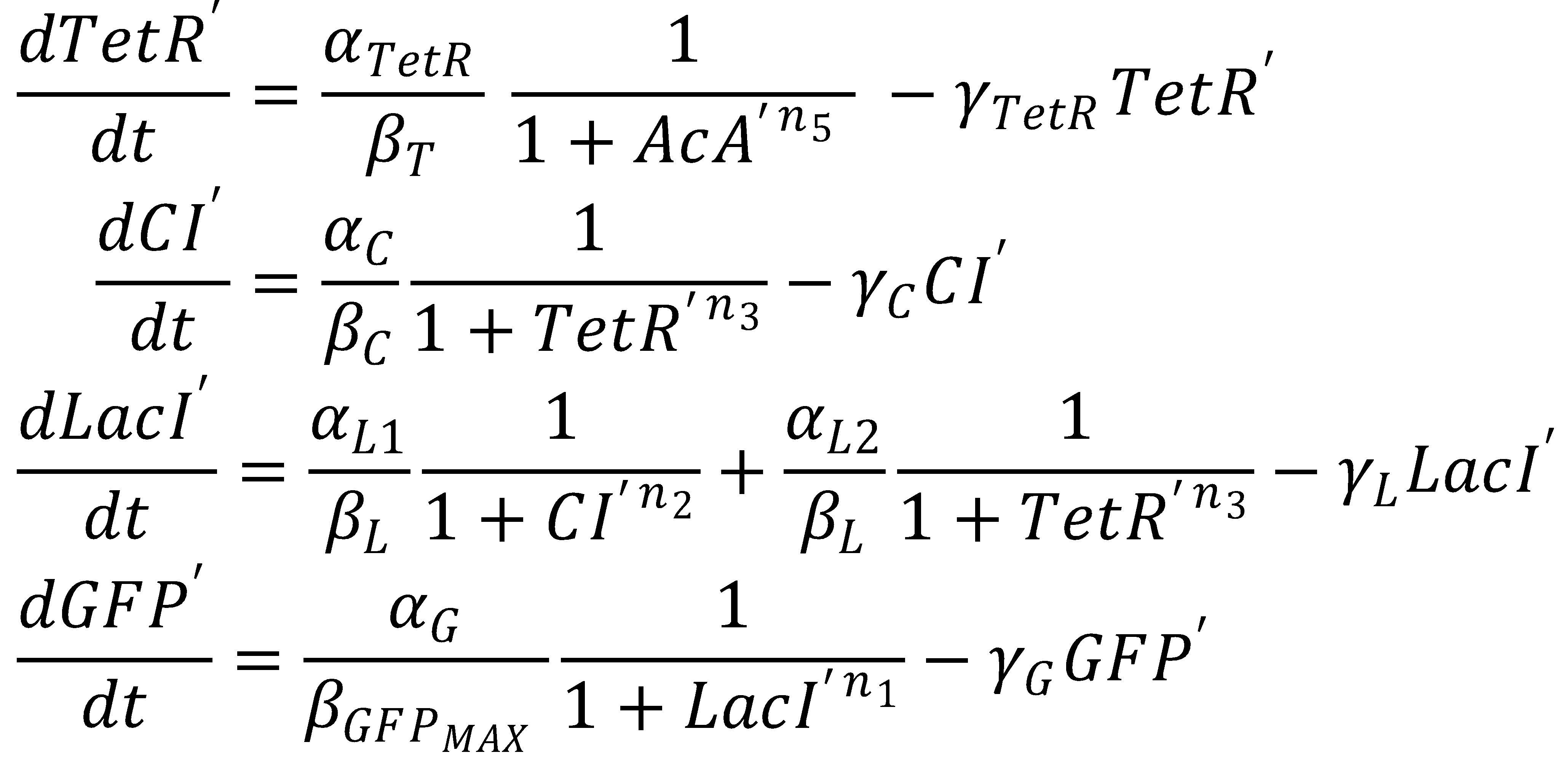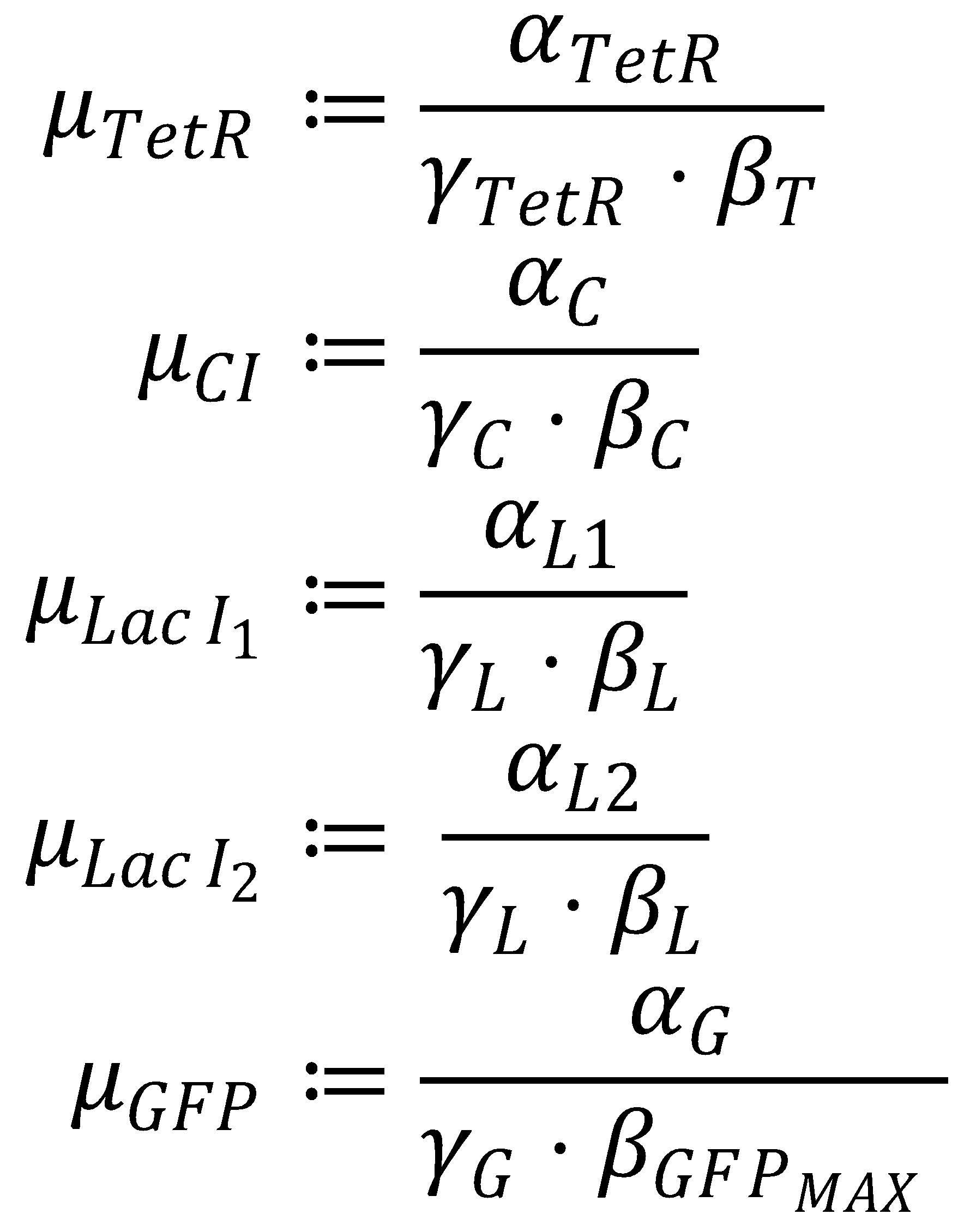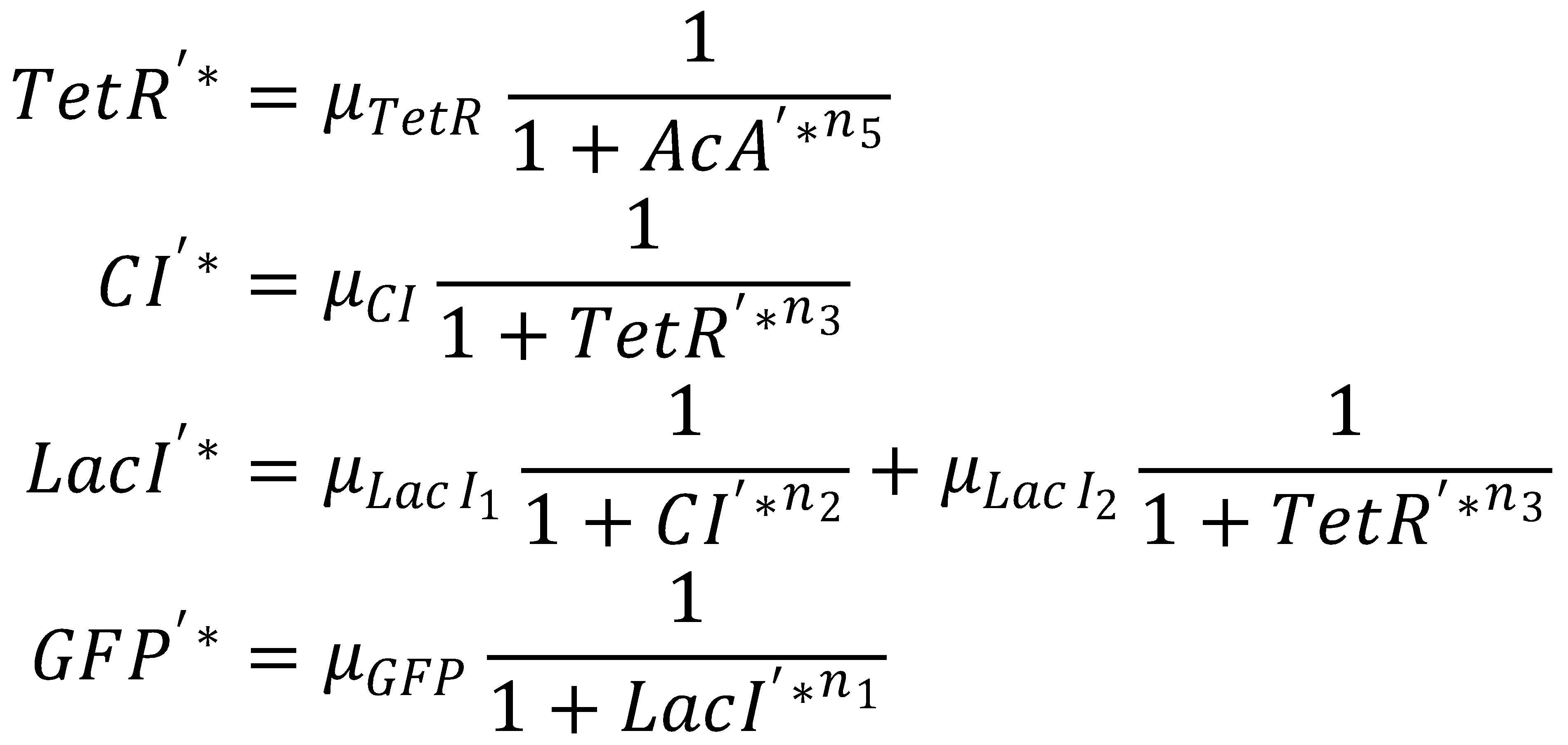Team:ETH Zurich/Modeling/Analytical Approximation
From 2011.igem.org
(→3. Partial Analytical Solution) |
(→3. Partial Analytical Solution) |
||
| Line 71: | Line 71: | ||
We can solve the ordinary differential equation we derived before by integrating wrt. to z twice, thereby introducing an unknown integration constant κ: | We can solve the ordinary differential equation we derived before by integrating wrt. to z twice, thereby introducing an unknown integration constant κ: | ||
[[File:ETHZ-KPP-Approx-ODE-PartialSol.png|365px|center|thumb|Parital solution with open parameter κ to the ordinary differential equation arising from the KPP equation.]] | [[File:ETHZ-KPP-Approx-ODE-PartialSol.png|365px|center|thumb|Parital solution with open parameter κ to the ordinary differential equation arising from the KPP equation.]] | ||
| + | To determine κ, we apply the dead-zone concept. | ||
== Dead Zone Concept == | == Dead Zone Concept == | ||
Revision as of 15:10, 28 October 2011

Contents |
Analytical Approximation
GFP Band: Dimensionless Model
1. Dimensionless Species
We nondimensionalized all species occurring in the equations for the GFP band circuit.
2. Dimensionless Equations
The dimensionless equations for the dynamics of the band-generating system then read
3. Steady State
4. Dimensionless Groupings
The equation system can be simplified by introducing the following dimensionless groupings:
This yields the final equations for steady state:
Steady State Gradient Approximation
We derived the gradient formation dynamics analytically already in the reaction-diffusion model. However, there we only solved the resulting partial differential equation numerically. Here, we are going to venture forth and give a suitable analytical approximation!
Kolmogorov-Petrovsky-Piskounov Equation
In mathematics, the problem posed by general reaction-diffusion systems form is known as the Kolmogorov-Petrovsky-Piskounov Equation. In the case of acetaldehyde as diffusing and reacting molecule, the equation has the following general structure:
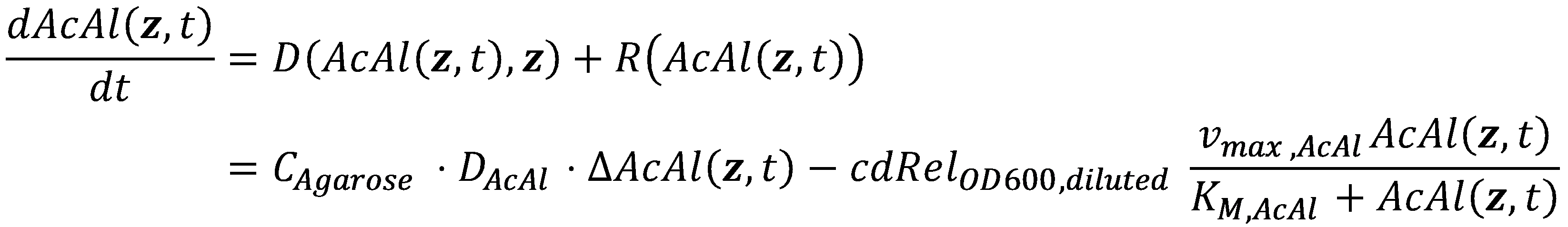
Boundary Conditions
For the analytical approximation of the acetaldehyde gradient, we only consider the boundary condition that the concentration at the reservoir (located at z = 0) stays constant:
This is in contrast to the full model, where we enforce that no diffusion occurs through the opening at the side of the channel which is closed and not connected to the reservoir. However, we will see that for our circuit, this assumption is valid as long as we are inside the detection range of our circuit, which is the case that we are interested in. In the end, we can also detect if we go out of detection range in the analytical model and make sure this assumption holds.
Solution at Steady State
At steady state, we have to solve the KPP equation for the time derivative set to zero, i.e.
1. Simplification
Before we get started with solving the actual equation, we simplify it by gathering all the parameters we can:
2. Approximation
Next, we assume that the Michaelis-Menten-type degradation in our KPP operates at or almost at saturation for the relevant part of the gradient. This assumption holds for inputs that are within detection range according to the exact numerical integration of the KPP equation.
This means we can approximate the reaction term around saturation, i.e. AcAl*(z) >> KM,AcAl:
We have now arrived at a solvable ordinary differential equation for the acetaldehyde gradient:
3. Partial Analytical Solution
We can solve the ordinary differential equation we derived before by integrating wrt. to z twice, thereby introducing an unknown integration constant κ:
To determine κ, we apply the dead-zone concept.
Dead Zone Concept
Analytical Solution
References
http://www.ncbi.nlm.nih.gov/pubmed/16209545
 "
"