Team:Bielefeld-Germany/Data Page
From 2011.igem.org
(Difference between revisions)
(→How our System works) |
|||
| Line 10: | Line 10: | ||
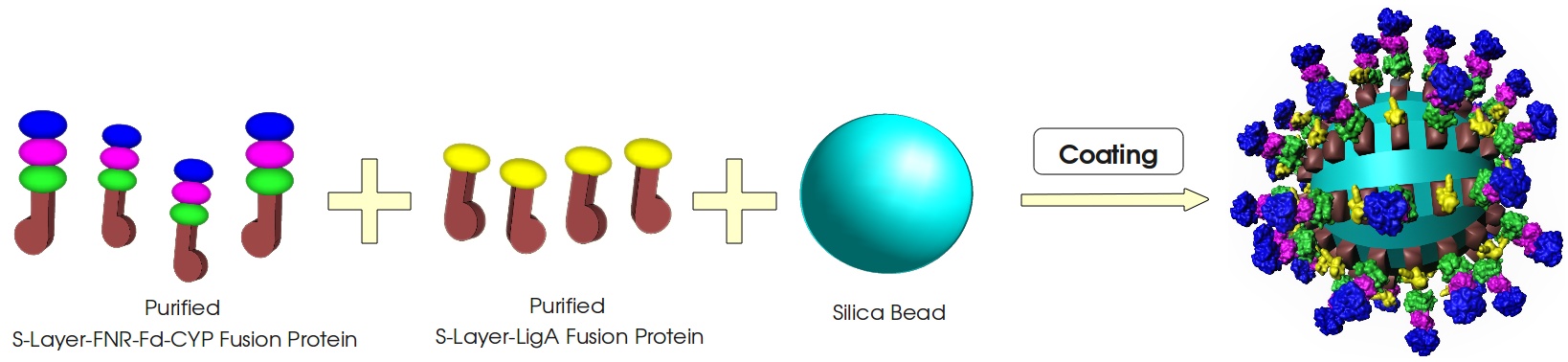
[[Image:Bielefeld_2011_Bead-Coating_v1.jpg|center|700px|thumb|'''Figure 2: Coating of the silica beads with the S-Layer fusion proteins.''' Every silica bead gets covered by a geometric S-Layer film consisting of an alternating structure of the two essential S-Layer fusion proteins.]] | [[Image:Bielefeld_2011_Bead-Coating_v1.jpg|center|700px|thumb|'''Figure 2: Coating of the silica beads with the S-Layer fusion proteins.''' Every silica bead gets covered by a geometric S-Layer film consisting of an alternating structure of the two essential S-Layer fusion proteins.]] | ||
| - | [[Image:IGEM_Bielefeld_Project.jpg|center|700px|thumb|'''Figure 3: Visualization of our cell-free Bisphenol A biosensor system with all essential components.''' Bisphenol A (BPA) is reduced by the electrons from NADH transferred by the ferredoxin-NADP+ oxidoreductase (FNR, <partinfo>BBa_K525499</partinfo>), ferredoxin (Fd, <partinfo>BBa_K123000</partinfo>) and cytochrome P450 (CYP, <partinfo>BBa_K123001</partinfo>) which are fused to a S-Layer protein. The molecular beacon (hairpin structure) binds two short DNA oligos. The NAD<sup>+</sup>-dependent ligase (LigA), which is also fused to a S-Layer, ligates the two oligos so that the hairpin structure opens up and the fluorophore is able to emit light after extinction.]] | + | [[Image:IGEM_Bielefeld_Project.jpg|center|700px|thumb|'''Figure 3: Visualization of our cell-free Bisphenol A biosensor system with all essential components.''' Bisphenol A (BPA) is reduced by the electrons from NADH transferred by the ferredoxin-NADP+ oxidoreductase (FNR, <partinfo>BBa_K525499</partinfo>), ferredoxin (Fd, <partinfo>BBa_K123000</partinfo>) and cytochrome P450 (CYP, <partinfo>BBa_K123001</partinfo>) which are fused to a S-Layer protein. The molecular beacon (hairpin structure) binds two short DNA oligos. The NAD<sup>+</sup>-dependent ligase (LigA), which is also fused to a S-Layer protein, ligates the two oligos so that the hairpin structure opens up and the fluorophore is able to emit light after extinction.]] |
==Data For Our Favorite New Parts== | ==Data For Our Favorite New Parts== | ||
Revision as of 14:11, 20 September 2011


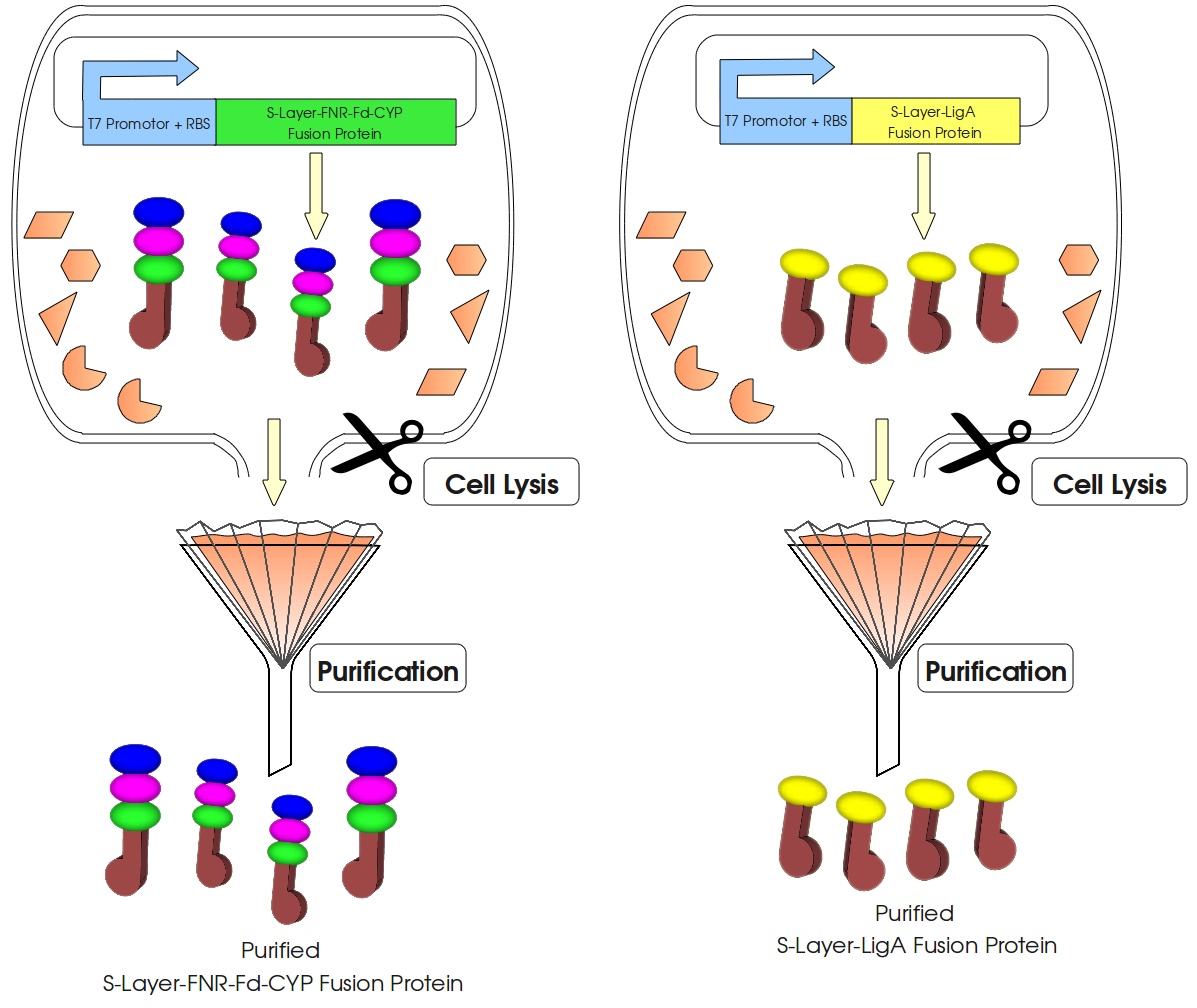
This page gives a basic overview about our cell-free Bisphenol A biosensor system and the BioBricks we have used. A more detailed description of the biosensor system can be found in our project description and in the Bisphenol A, S-Layer and NAD+ detection background subsections.
Contents |
How our System works
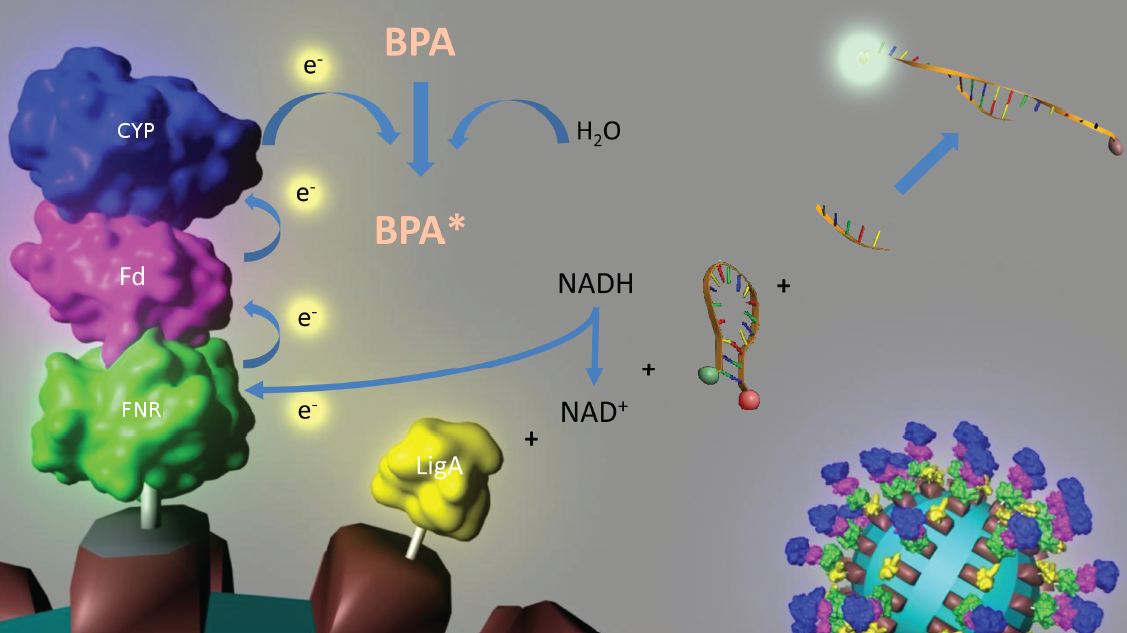
Figure 3: Visualization of our cell-free Bisphenol A biosensor system with all essential components. Bisphenol A (BPA) is reduced by the electrons from NADH transferred by the ferredoxin-NADP+ oxidoreductase (FNR, <partinfo>BBa_K525499</partinfo>), ferredoxin (Fd, <partinfo>BBa_K123000</partinfo>) and cytochrome P450 (CYP, <partinfo>BBa_K123001</partinfo>) which are fused to a S-Layer protein. The molecular beacon (hairpin structure) binds two short DNA oligos. The NAD+-dependent ligase (LigA), which is also fused to a S-Layer protein, ligates the two oligos so that the hairpin structure opens up and the fluorophore is able to emit light after extinction.
Data For Our Favorite New Parts
- [http://partsregistry.org/Part:BBa_K525305 Main Page] - Fusion Protein of S-Layer SgsE and mCitrine: This fluorescent S-layer fusion protein is used to characterize purification methods and to demonstrate the S-layer's ability to self-assemble on surfaces.
- [http://partsregistry.org/Part:BBa_K525517 Main Page] - Fusion Protein of BisdA and BisdB (expressed): This fusion protein improves the bisphenol A degradation in E. coli compared to the so far in the partsregistry existing BPA degrading BioBricks.
- [http://partsregistry.org/Part:BBa_K525710 Main Page] - NAD+-dependent DNA ligase from E. coli : This enzyme enables determination of NAD+ even in very low concentrations by coupling it with a molecular beacon based assay.
Data For Pre-existing Parts
- [http://partsregistry.org/Part:BBa_K123000:Experience Experience] - BisdA degrades Bisphenol A when used with BisdB, BBa_K123000 (University of Alberta, iGEM 2008): Complete degradation of 120 mg L-1 Bisphenol A with polycistronic bisdAB gene in 30-33 h. Even faster (21-24 h) when using a fusion protein of BisdA and BisdB.
- [http://partsregistry.org/Part:BBa_K123001:Experience Experience] - BisdB degrades Bisphenol A when used with BisdA, BBa_K123001 (University of Alberta, iGEM 2008): Complete degradation of 120 mg L-1 Bisphenol A with polycistronic bisdAB gene in 30-33 h. Even faster (21-24 h) when using a fusion protein of BisdA and BisdB.
We've Also Characterized the Following Parts
- [http://partsregistry.org/Part:BBa_K525405 Main Page] - Fusion Protein of S-Layer SbpA and mCitrine: This fluorescent S-layer fusion protein is used to characterize purification methods and to demonstrate the S-layer's ability to self-assemble on surfaces.
- [http://partsregistry.org/Part:BBa_K525512 Main Page] - Polycistronic expression of BisdA and BisdB: This is the version of BPA degrading BioBricks found in the partsregistry - comparison to our fusion protein <partinfo>K525515</partinfo>.
 "
"