Team:Paris Bettencourt/Designs
From 2011.igem.org
| Line 1: | Line 1: | ||
{{:Team:Paris_Bettencourt/tpl_test}} | {{:Team:Paris_Bettencourt/tpl_test}} | ||
<html> | <html> | ||
| + | <h1>Designs overview</h1> | ||
<h2>Introduction</h2> | <h2>Introduction</h2> | ||
| - | <p> | + | <p>Our goal for this summer is characterizing the exchange of proteins through nanotubes as best as we can. For this we do have two different expetiments. First we rely on <em>simple transfer trough the nanotubes</em> without having any kind of feedback. We then try to use <em>signal amplification</em> to enhance our results and give new evidences for a non-specific cell-to-cell communication channel.</p> |
| + | <p>All of the designs below have not been tried for several reasons. Sometimes the cloning steps are too hazardous, sometimes we missed a key component. We concentrated our effort this summer on the <a href="https://2011.igem.org/Team:Paris_Bettencourt/T7_diffusion">T7 RNA polymerase diffusion</a>, the <a href="https://2011.igem.org/Team:Paris_Bettencourt/ComS_diffusion">ComS diffusion</a> and the <a href="https://2011.igem.org/Team:Paris_Bettencourt/tRNA_diffusion">tRNA amber diffusion</a>.</p> | ||
| + | <p>We had neither direct access to an electronic microscope nor the time to do any electronic microscopy. Our experiments therefore can only support the existence of a non-specific cell-to-cell communication channel which is strongly suspected to be the nanotubes described by Ben-Yehuda.</p> | ||
<br> | <br> | ||
| Line 10: | Line 13: | ||
<br> | <br> | ||
| - | < | + | <h2>Simple transfer</h2> |
<div style="float:right; width: 250px; margin: 15px;"><a href="https://2011.igem.org/File:Nanotube_and_GFP.jpeg"><img src="../wiki/images/e/ed/Nanotube_and_GFP.jpeg" style="width:100%;"></a> | <div style="float:right; width: 250px; margin: 15px;"><a href="https://2011.igem.org/File:Nanotube_and_GFP.jpeg"><img src="../wiki/images/e/ed/Nanotube_and_GFP.jpeg" style="width:100%;"></a> | ||
| Line 16: | Line 19: | ||
| - | <p>In the paper from Dubey and Ben Yehuda, many types of experiments | + | <p>In the paper from Dubey and Ben Yehuda, many types of experiments were done to demonstrate the existence and the efficiency of the nanotube network as a mean of communication between bacteria. We wanted to do again some experiments. We wanted to be sure we are able to put ourself in the conditions where the nanotubes existt in our own laboratory. We also tried to reproduce results of the article that are the object of many discussions in the community.</p> |
| - | <p>In these experiments, there are no amplification system. We simply obeserve, directly | + | <p>In these experiments, there are no amplification system. We simply obeserve, directly or indirectly the molecules that have passed through the tubes.</p> |
| - | <p> | + | <p>Experimental design for this step are the following:</p> |
<ul> | <ul> | ||
<li><a href="https://2011.igem.org/Team:Paris_Bettencourt/GFP_diff">The GFP diffusion experiment:</a> This experiment is the keystone experiment of the paper. One strain expressing GFP is mixed with one wild type strain, and the GFP proteins are diffusing from the GFP cells to the colorless cells.</li> | <li><a href="https://2011.igem.org/Team:Paris_Bettencourt/GFP_diff">The GFP diffusion experiment:</a> This experiment is the keystone experiment of the paper. One strain expressing GFP is mixed with one wild type strain, and the GFP proteins are diffusing from the GFP cells to the colorless cells.</li> | ||
| - | <li><a href="https://2011.igem.org/Team:Paris_Bettencourt/GFPLac_diffusion">The YFP-TetR/TetO array experiment:</a> This experiment is an improvement of the previous one. To observe significant fluorescence in the neighboring cell, lots of molecule have to pass through the tubes. Using the affinity of the TetR for the TetO array, we want to concentrate at one point the | + | <li><a href="https://2011.igem.org/Team:Paris_Bettencourt/GFPLac_diffusion">The YFP-TetR/TetO array experiment:</a> This experiment is an improvement of the previous one. To observe significant fluorescence in the neighboring cell, lots of molecule have to pass through the tubes. Using the affinity of the TetR for the TetO array, we want to concentrate at one point the YFP molecules to see them better.</li> |
| - | <li><a href="https://2011.igem.org/Team:Paris_Bettencourt/Atb_exp">The antibiotics resistance exchange experiment:</a> This experiment is the most controversed experiment of the paper. The | + | <li><a href="https://2011.igem.org/Team:Paris_Bettencourt/Atb_exp">The antibiotics resistance exchange experiment:</a> This experiment is the most controversed experiment of the paper. The authors claim that a mix of two strains, each of them holding a resistance for a given antibiotic can survive together in a medium containig the two antibiotics because they exchange resistance enzymes through the nanotubes. We found that there are other possible explanations for this experiment than the existence of the nanotubes.</li> |
</ul> | </ul> | ||
<br> | <br> | ||
| - | < | + | <h2>Designs for nanotube characterization</h2> |
| - | < | + | <h3>Key specification of our designs</h3> |
| - | <p>The aim of our project is | + | <p>The aim of our project is to <em>characterize</em> and try to control the <em>cellular communication through the nanotubes</em> by using the techniques of synthetic biology. At its beginnings, synthetic biology was not an engineering field. The idea was to <em>build artificial systems to explore more deeply the properties of cells</em>. Our project is clearly following this idea.</p> |
| - | <p>The first question about | + | <p>The first question about nanotubes we asked ourself is <em>what kind of molecules can we pass through the nanotubes?</em> Do they have to be big? Do they have to be very small? The paper shows that GFP, calcein and plasmids can pass, but what if the protein is bigger?</p> |
| - | <p> | + | <p>We then wondered at <em>the transfer process in the tube</em>. Is it simple diffusion? Is there an active process?</p> |
| - | <p>We started designing the project these two questions in mind. | + | <p>We started designing the project with these two questions in mind. Using several molecules of <em>very different sizes</em>, from a T7 polymerase to a tRNA amber supressor, we aim to test the capability and size limit for the molecules that can pass through the tubes. But, if the diffusion is passive, the speed of diffusion should be proportional to the diffusion coefficient D, that is to say invertially proportinal to the radius of the object. Can we design a fast-responding system that would allow us to measure this time?</p> |
| - | </p> | + | </p>We decided that our constructs should follow the global idea summed up in the following picture:</p> |
| Line 49: | Line 52: | ||
| - | <p>The problem is that each design needs a different couple of signal emittor/receptor. | + | <p>The problem is that each design needs a different couple of signal emittor/receptor. However, even with good modeling, we can not easily predict the response time because we do not know much about transfer through the nanotubes. We tried to mitigate the impact of this issue:</p> |
<p> | <p> | ||
<ul> | <ul> | ||
| - | <li> | + | <li>We try to use each time the same actuator and the same monitor to have comparable response times in different systems.</li> |
| - | <li> | + | <li>The relevant measure is not response time itself but the increase of response time between "all in one cell" strains (where all of the construct emitter and receptor is in one strain) and the emitter strain/receptor strain mix (where the two halves of the construct are not in the same strains).</li> |
</ul> | </ul> | ||
</p> | </p> | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
<br> | <br> | ||
| - | < | + | <h3>Designs testing B. Subtilis to B. Subtilis communication</h3> |
| - | <p>In B. | + | <p>In <i>B.subtilis</i>, nanotubes are reported to connect directly the two cytoplasms. We had to find a molecule that can be expressed in the first one and trigger a genetic circuit in the second one. The idea is summed up on this image:</p> |
<a href="https://2011.igem.org/File:cytoplasm_cytoplasm_communication.jpg"><img src="../wiki/images/4/49/Cytoplasm_cytoplasm_communication.jpg" style="width: 500px; margin-left: 185px;"></a></br> | <a href="https://2011.igem.org/File:cytoplasm_cytoplasm_communication.jpg"><img src="../wiki/images/4/49/Cytoplasm_cytoplasm_communication.jpg" style="width: 500px; margin-left: 185px;"></a></br> | ||
<p><center><b><u>Fig2:</u> Schematic of the supposed connection between the cells via the nanotubes</b></center></p> | <p><center><b><u>Fig2:</u> Schematic of the supposed connection between the cells via the nanotubes</b></center></p> | ||
| - | + | <p>As we said earlier, the goal is to pass several types of molecules, with different sizes, and see if we can observe an increase in the response time with the size of the molecule. The molecules chosen cover 3 order of magnitude of size. They are there classified from the biggest to the smallest.</p> | |
| - | <p>As we said earlier, the | + | |
<ul> | <ul> | ||
| - | <li><a href="https://2011.igem.org/Team:Paris_Bettencourt/T7_diffusion">The T7 polymerase diffusion:</a> The T7 RNA polymerase is the RNA polymerase of the T7 phage. This is a pretty big molecule, that | + | <li><a href="https://2011.igem.org/Team:Paris_Bettencourt/T7_diffusion">The T7 polymerase diffusion:</a> The T7 RNA polymerase is the RNA polymerase of the T7 phage. This is a pretty big molecule, that recognizes a very specific promoter orthogonal to <i>B.subtilis</i>.</li> |
| - | <li><a href="https://2011.igem.org/Team:Paris_Bettencourt/KinA_diff">The KinA diffusion:</a> The idea is to make a complex partner to diffuse from one cell to the other in order to trigger the sporulation of the second one, even in | + | <li><a href="https://2011.igem.org/Team:Paris_Bettencourt/KinA_diff">The KinA diffusion:</a> The idea is to make a complex partner to diffuse from one cell to the other in order to trigger the sporulation of the second one, even in exponential phase.</li> |
| - | <li><a href="https://2011.igem.org/Team:Paris_Bettencourt/ComS_diffusion">The ComS diffusion:</a>ComS is an inhibitor of the MecA protease in B. | + | <li><a href="https://2011.igem.org/Team:Paris_Bettencourt/ComS_diffusion">The ComS diffusion:</a> ComS is an inhibitor of the MecA protease in <i>B.subtilis</i>. It plays a key role in the triggering of the competence and sporulation mechanisms. The idea is to trigger the switch of the MeKS system of the receptor cell by diffusing ComS through the nanotubes. We also need to prevent the first cell from sporulating.</li> |
| - | <li><a href="https://2011.igem.org/Team:Paris_Bettencourt/tRNA_diffusion">The amber suppressor tRNA diffusion:</a> The principle of this design is to produce in one cell a amber supressor tRNA that will diffuse through the nanotubes. The receptor cell holds the gene for a T7 with amber stop codons that cannot be translated into a functional protein as long as the tRNA amber suppressor is not present in the cell. Once expressed, the | + | <li><a href="https://2011.igem.org/Team:Paris_Bettencourt/tRNA_diffusion">The amber suppressor tRNA diffusion:</a> The principle of this design is to produce in one cell a amber supressor tRNA that will diffuse through the nanotubes. The receptor cell holds the gene for a T7 with amber stop codons that cannot be translated into a functional protein as long as the tRNA amber suppressor is not present in the cell. Once expressed, the functional amber T7 RNA polymerase will trigger the T7 amplification system.</li> |
</ul> | </ul> | ||
<br> | <br> | ||
| - | < | + | <br/> |
| + | |||
| + | <h3>Inter-species communication</h3> | ||
| + | |||
| + | <p>The article demonstrates that nanotubes established inside the <i>B.subtilis</i> familly, but can also be established between <i>B.subtilis</i> and an <i>E.coli</i>. Theses two types of bacteria are really different since one is Gramm+ and the other is Gramm-. Their membrane is really different, since there is no periplasm in <i>B.subtilis</i>. The existence of <em>inter-species nanotubes</em> did really spiked our interest and we wanted also to create designs to test this aspect of the communication.</p> | ||
| + | |||
| + | <p><i>B.subtilis</i> is Gramm-. The nanotubes seem to create a jonction from the cytoplasm of the first cell to the cytoplasm of the second one. In the case of the Gramm+ bacteria, <i>like E.coli</i>, we wonder which membrane nanotubes target. Do they create a link with the periplasm, or directly link the two cells cytoplasm? To test the two hypotheses, we tried to create a design for each of the two possibilities. If one work and the other not, we will know the answer to this question.</p> | ||
| + | |||
| + | <h4>Designs for B. Subtilis-E. Coli communication</h4> | ||
| - | <p>As we said above, if | + | <p>As we said above, if nanotube communication can be established between <i>E.coli</i> and <i>B.subtilis</i>, we do not know if the communication happens throught the cytoplasm or the periplasm. To test these two hypotheses, we have two types of designs:</p> |
| - | < | + | <h4>If communication happens with the cytoplasm</h4> |
<p>The cheme below summ up the kind of connection that can be established if the nanotube connect the two cytoplasm. Hence, some of the design we shown above are still available from B. Subtilis to E. Coli.</p> | <p>The cheme below summ up the kind of connection that can be established if the nanotube connect the two cytoplasm. Hence, some of the design we shown above are still available from B. Subtilis to E. Coli.</p> | ||
Revision as of 13:33, 17 September 2011

Designs overview
Introduction
Our goal for this summer is characterizing the exchange of proteins through nanotubes as best as we can. For this we do have two different expetiments. First we rely on simple transfer trough the nanotubes without having any kind of feedback. We then try to use signal amplification to enhance our results and give new evidences for a non-specific cell-to-cell communication channel.
All of the designs below have not been tried for several reasons. Sometimes the cloning steps are too hazardous, sometimes we missed a key component. We concentrated our effort this summer on the T7 RNA polymerase diffusion, the ComS diffusion and the tRNA amber diffusion.
We had neither direct access to an electronic microscope nor the time to do any electronic microscopy. Our experiments therefore can only support the existence of a non-specific cell-to-cell communication channel which is strongly suspected to be the nanotubes described by Ben-Yehuda.
Simple transfer
In the paper from Dubey and Ben Yehuda, many types of experiments were done to demonstrate the existence and the efficiency of the nanotube network as a mean of communication between bacteria. We wanted to do again some experiments. We wanted to be sure we are able to put ourself in the conditions where the nanotubes existt in our own laboratory. We also tried to reproduce results of the article that are the object of many discussions in the community.
In these experiments, there are no amplification system. We simply obeserve, directly or indirectly the molecules that have passed through the tubes.
Experimental design for this step are the following:
- The GFP diffusion experiment: This experiment is the keystone experiment of the paper. One strain expressing GFP is mixed with one wild type strain, and the GFP proteins are diffusing from the GFP cells to the colorless cells.
- The YFP-TetR/TetO array experiment: This experiment is an improvement of the previous one. To observe significant fluorescence in the neighboring cell, lots of molecule have to pass through the tubes. Using the affinity of the TetR for the TetO array, we want to concentrate at one point the YFP molecules to see them better.
- The antibiotics resistance exchange experiment: This experiment is the most controversed experiment of the paper. The authors claim that a mix of two strains, each of them holding a resistance for a given antibiotic can survive together in a medium containig the two antibiotics because they exchange resistance enzymes through the nanotubes. We found that there are other possible explanations for this experiment than the existence of the nanotubes.
Designs for nanotube characterization
Key specification of our designs
The aim of our project is to characterize and try to control the cellular communication through the nanotubes by using the techniques of synthetic biology. At its beginnings, synthetic biology was not an engineering field. The idea was to build artificial systems to explore more deeply the properties of cells. Our project is clearly following this idea.
The first question about nanotubes we asked ourself is what kind of molecules can we pass through the nanotubes? Do they have to be big? Do they have to be very small? The paper shows that GFP, calcein and plasmids can pass, but what if the protein is bigger?
We then wondered at the transfer process in the tube. Is it simple diffusion? Is there an active process?
We started designing the project with these two questions in mind. Using several molecules of very different sizes, from a T7 polymerase to a tRNA amber supressor, we aim to test the capability and size limit for the molecules that can pass through the tubes. But, if the diffusion is passive, the speed of diffusion should be proportional to the diffusion coefficient D, that is to say invertially proportinal to the radius of the object. Can we design a fast-responding system that would allow us to measure this time?
We decided that our constructs should follow the global idea summed up in the following picture: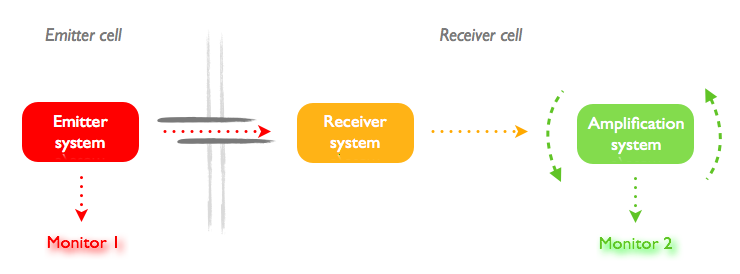
The problem is that each design needs a different couple of signal emittor/receptor. However, even with good modeling, we can not easily predict the response time because we do not know much about transfer through the nanotubes. We tried to mitigate the impact of this issue:
- We try to use each time the same actuator and the same monitor to have comparable response times in different systems.
- The relevant measure is not response time itself but the increase of response time between "all in one cell" strains (where all of the construct emitter and receptor is in one strain) and the emitter strain/receptor strain mix (where the two halves of the construct are not in the same strains).
Designs testing B. Subtilis to B. Subtilis communication
In B.subtilis, nanotubes are reported to connect directly the two cytoplasms. We had to find a molecule that can be expressed in the first one and trigger a genetic circuit in the second one. The idea is summed up on this image:
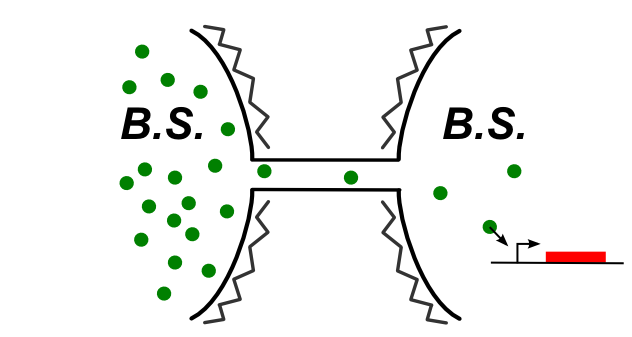
As we said earlier, the goal is to pass several types of molecules, with different sizes, and see if we can observe an increase in the response time with the size of the molecule. The molecules chosen cover 3 order of magnitude of size. They are there classified from the biggest to the smallest.
- The T7 polymerase diffusion: The T7 RNA polymerase is the RNA polymerase of the T7 phage. This is a pretty big molecule, that recognizes a very specific promoter orthogonal to B.subtilis.
- The KinA diffusion: The idea is to make a complex partner to diffuse from one cell to the other in order to trigger the sporulation of the second one, even in exponential phase.
- The ComS diffusion: ComS is an inhibitor of the MecA protease in B.subtilis. It plays a key role in the triggering of the competence and sporulation mechanisms. The idea is to trigger the switch of the MeKS system of the receptor cell by diffusing ComS through the nanotubes. We also need to prevent the first cell from sporulating.
- The amber suppressor tRNA diffusion: The principle of this design is to produce in one cell a amber supressor tRNA that will diffuse through the nanotubes. The receptor cell holds the gene for a T7 with amber stop codons that cannot be translated into a functional protein as long as the tRNA amber suppressor is not present in the cell. Once expressed, the functional amber T7 RNA polymerase will trigger the T7 amplification system.
Inter-species communication
The article demonstrates that nanotubes established inside the B.subtilis familly, but can also be established between B.subtilis and an E.coli. Theses two types of bacteria are really different since one is Gramm+ and the other is Gramm-. Their membrane is really different, since there is no periplasm in B.subtilis. The existence of inter-species nanotubes did really spiked our interest and we wanted also to create designs to test this aspect of the communication.
B.subtilis is Gramm-. The nanotubes seem to create a jonction from the cytoplasm of the first cell to the cytoplasm of the second one. In the case of the Gramm+ bacteria, like E.coli, we wonder which membrane nanotubes target. Do they create a link with the periplasm, or directly link the two cells cytoplasm? To test the two hypotheses, we tried to create a design for each of the two possibilities. If one work and the other not, we will know the answer to this question.
Designs for B. Subtilis-E. Coli communication
As we said above, if nanotube communication can be established between E.coli and B.subtilis, we do not know if the communication happens throught the cytoplasm or the periplasm. To test these two hypotheses, we have two types of designs:
If communication happens with the cytoplasm
The cheme below summ up the kind of connection that can be established if the nanotube connect the two cytoplasm. Hence, some of the design we shown above are still available from B. Subtilis to E. Coli.
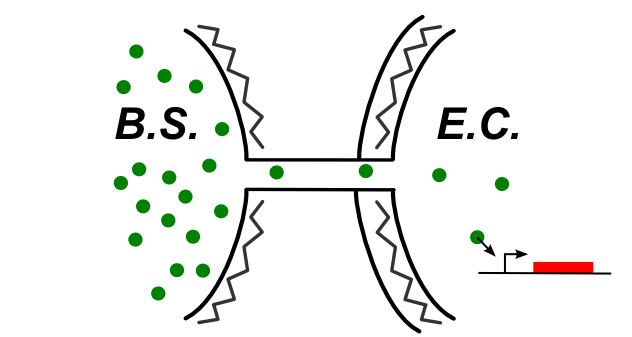
Here are the designs tested in this case:
- The T7 polymerase diffusion: The T7 RNA polymerase is the RNA polymerase of the T7 phage. This is a pretty big molecule, that recognize a very specific propoter orthogonal to the bacteria one.
- The C1(ind) diffusion: Thanks to our collaboration with the Pekin iGEM Team, we had the possibility to reuse the push-on push-off system they designed last year. The idea is to diffuse from subtilis the molecule C1 that will trigger a change in the toggle switch state
- The Xis protein diffusion: Xis is a small partner of an exisase. The latter will exise a stop codon on the DNA strand that is preventing the expression of the GFP.We had no time to go on with this design, but the idea is summed up on the linked page.
- The amber suppressor tRNA diffusion: The principle of this design is to produce in one cell a amber supressor tRNA that will diffuse through the nanotubes. The receptor cell holds the gene for a T7 with amber stop codons that cannot be translated into a functional protein as long as the tRNA amber suppressor is not present in the cell. Once expressed, the T7amber will trigger the T7 amplification system.
If communication happens with the periplasm
In case the communication happens with the periplasm, we have to think about molecules that can be then transported into the cytoplasm.

We have to think about more sophisticated approaches. The designs are as following:
- The MBP diffusion: We need a CRP+, MBP- E.Coli mutant. We produce the MBP protein in Bacillus subtilis and make it diffuse through the nanotubes. As long as the MBP has not reached the E. Coli periplasm, the cell cannot digest the maltose in the medium. The indirect induction of MalR by MBP triggers the expression of the GFP reporter.
- The OmpR diffusion: We need a OmpR- Receptor* E. Coli mutant. We produce the OmpR protein in Bacillus Subtilis. As long as the OmpR has not diffused from B. Subtilis, the signaling cascade cannot be activated. With the rescue by Bacillus Subtilis of the OmpR protein, the expression of the reporter gene is activated.
Can we conclude on these design?
If one of these designs works, we would prove that the interspecial nanotube communication exists and would find the eventual location of the connection with the membrane.
Designs for a Master-Slave system
The principle of Step 2 is to build a Master-Slave system, where the Master controls the state of the Slave cell in a monodirectional exchange.

Such a design implies the reversibility of all the sub-systems, the activators and the amplifiers in particular.
Several potential designs are summed-up below:
- The ComS system: The characterisation step of the MeKS system turn to be reversible. It can be considered as well as a Master/Slave system.
 "
"