Team:EPF-Lausanne/Our Project/T7 promoter variants
From 2011.igem.org
Lysis Selection System
Skip straight to: |
- Platereader Lysis Experiment: Using a platereader
- DNA Recovery Experiment: Recovering DNA after lysis
- DNA Selection Experiment: Selecting the right DNA
- T7 Promoter Variants: Making and Characterizing of T7 promoter variants for Lysis
DNA Recovery with Lysis
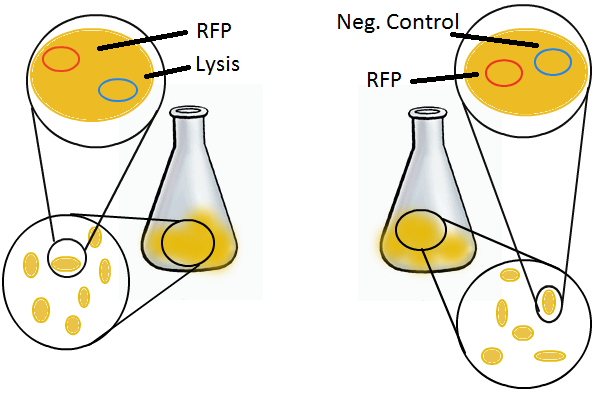
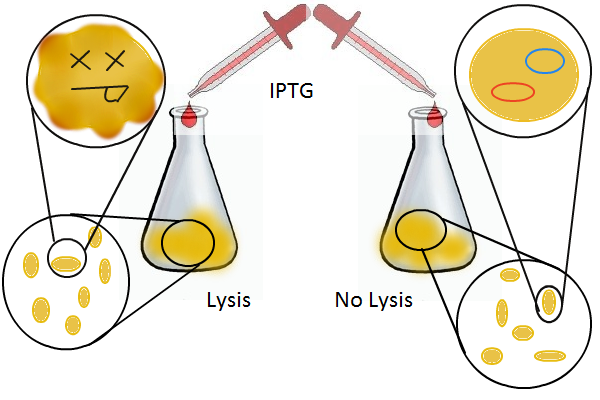
Having a lysis cassette driven by a T7 promoter is an important step towards being able to recover the DNA sequences of transcription factor and promoter mutants that have strong mutual affinities. To verify that basic lysing can be induced, we use the same IPTG experiment as for RFP, using both large samples for visual, qualitative confirmation and small samples in a platereader for quantitative, numerical confirmation.
Once the basic mechanism of cell lysing is confirmed, the next step is to show that DNA can be recovered from the supernatant. We grow two large cultures of cells. One contains cells that will lyse and release plasmids into the supernatant while the other has non-lysing, "normal" cells.
Adding IPTG to both flasks induces lysis in one set of cells but not in the others.
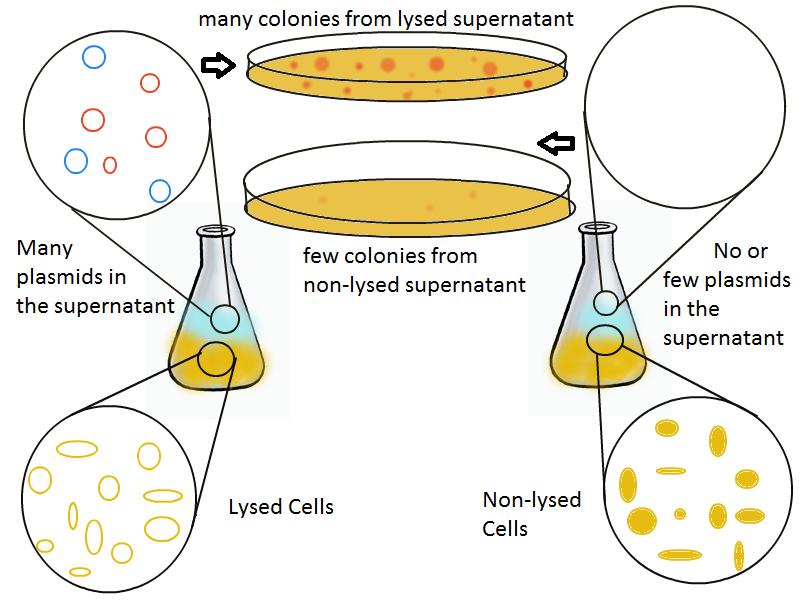
Thanks to qPCR, the supernatant harvested from the lysing culture reveals increased numbers of plasmids while the non-lysing culture exhibits significantly lower numbers of plasmids. A similar test involves transforming the supernatant (whose plasmid content is not known a priori) into cells and counting the number of resulting colonies. The plates containing transformations from the lysing supernatant have vastly superior number of colonies compared to the non-lysing supernatant transformations.
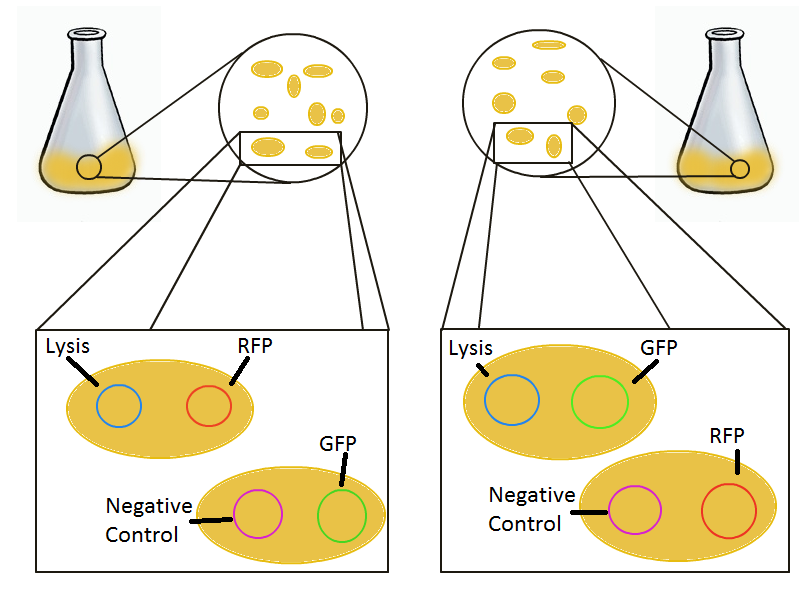
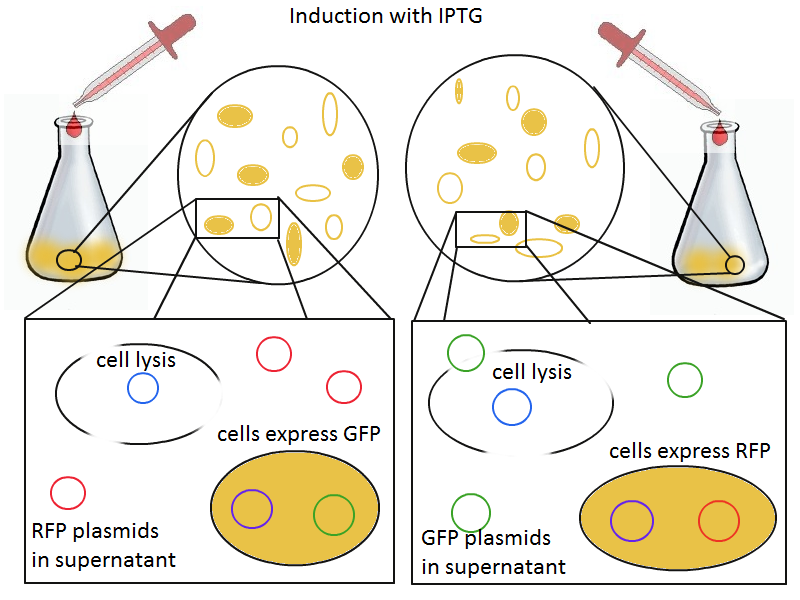
To round out the experiments for DNA recovery, it is essential that we show that the recovered DNA is in fact plasmid DNA from the relevant lysed cells. To that end, we again set up an experiment involving two flasks but this time each flask contains two cultures. For one flask, one culture is a co-transformation of a lysis plasmid and a RFP-containing plasmid and the other is a co-transformation of a negative control plasmid with a GFP-containing plasmid. In the other flask, the reverse is true: lysis is with GFP and negative control is with RFP.
Induction with IPTG lyses the cells with the lysis cassette but leaves the cells with the negative control free to produce the red fluorescence. Collecting the supernatant and sterile filtering it reveals, by qPCR or by transforming it and counting the colonies, that the supernatant contains RFP plasmids in one flask and GFP plasmids in the other. Meanwhile, the large flask from which the supernatant samples are taken should be yellow-green and pink respectively, since the remaining cells will fluoresce according to those colors.
 "
"