Yeast Toolkit Results
Yeast Toolkit Results
Promoter/Terminator Library
We biobricked 66% of the promoters and terminators that we set out to build. This will improve the ability of future igem teams that will use yeast to perform synthetic biology. We have successfully transformed a number of promoters into yeast(Bap2p, Tdh3p, Kre9p, Hh01p, Pry1p) in order to control expression of a GFP gene. The promoters are ready for flow-cytometry analysis.
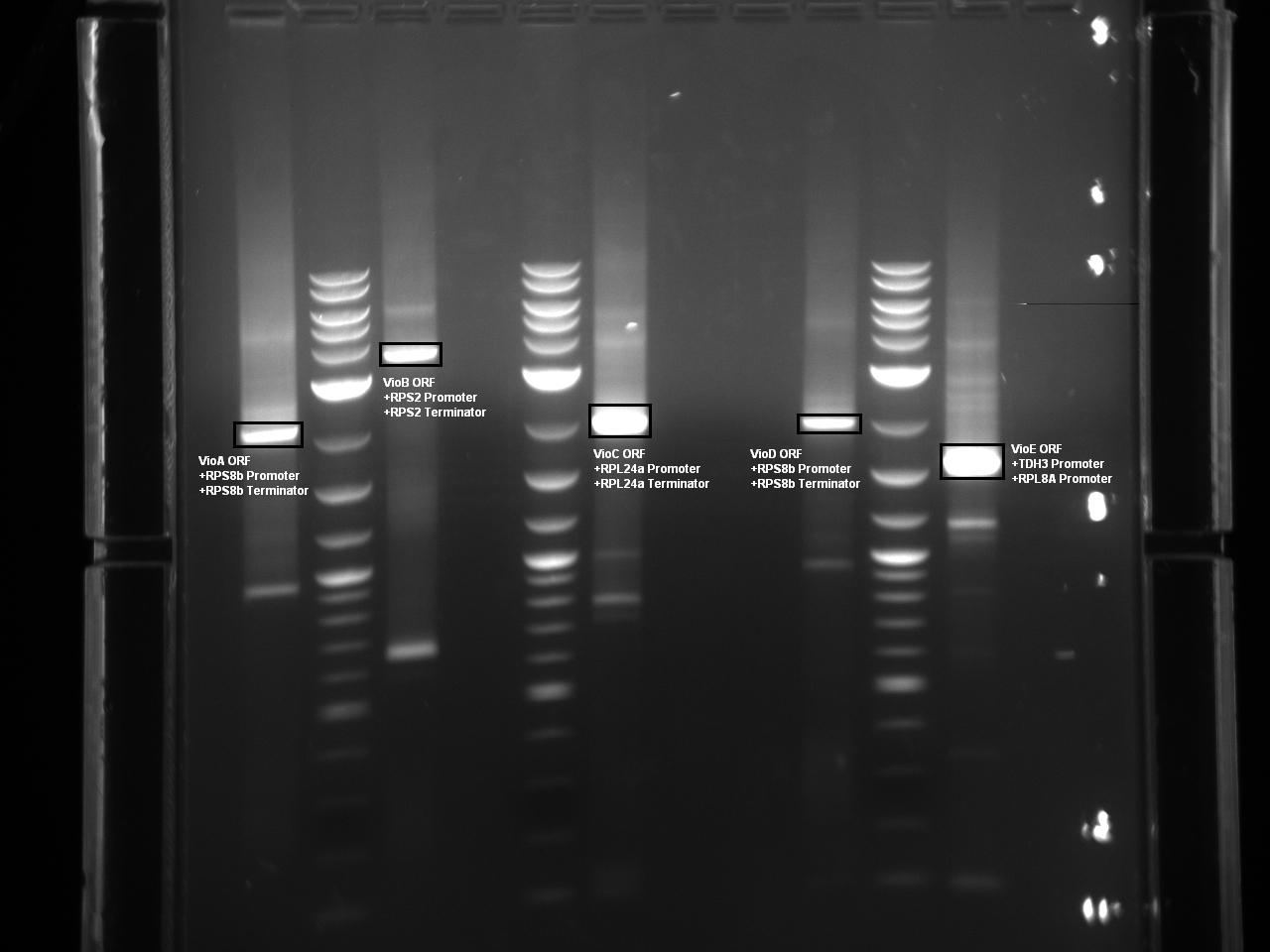
Five Violacein Expression Cassettes Ready for Vector Ligation and Yeast Transformation
At this point, Golden Gate Assembly compatible BsaI sites have been added to the 5 necessary violacein biosythesis expression cassettes via overhang PCR. The Herculase Fusion II polymerase was used in the reaction to lower the probability of point mutations. The following Gel shows each expression cassette digested with BsaI prior to gel extraction and purification.
Violacein loxPsym Integration
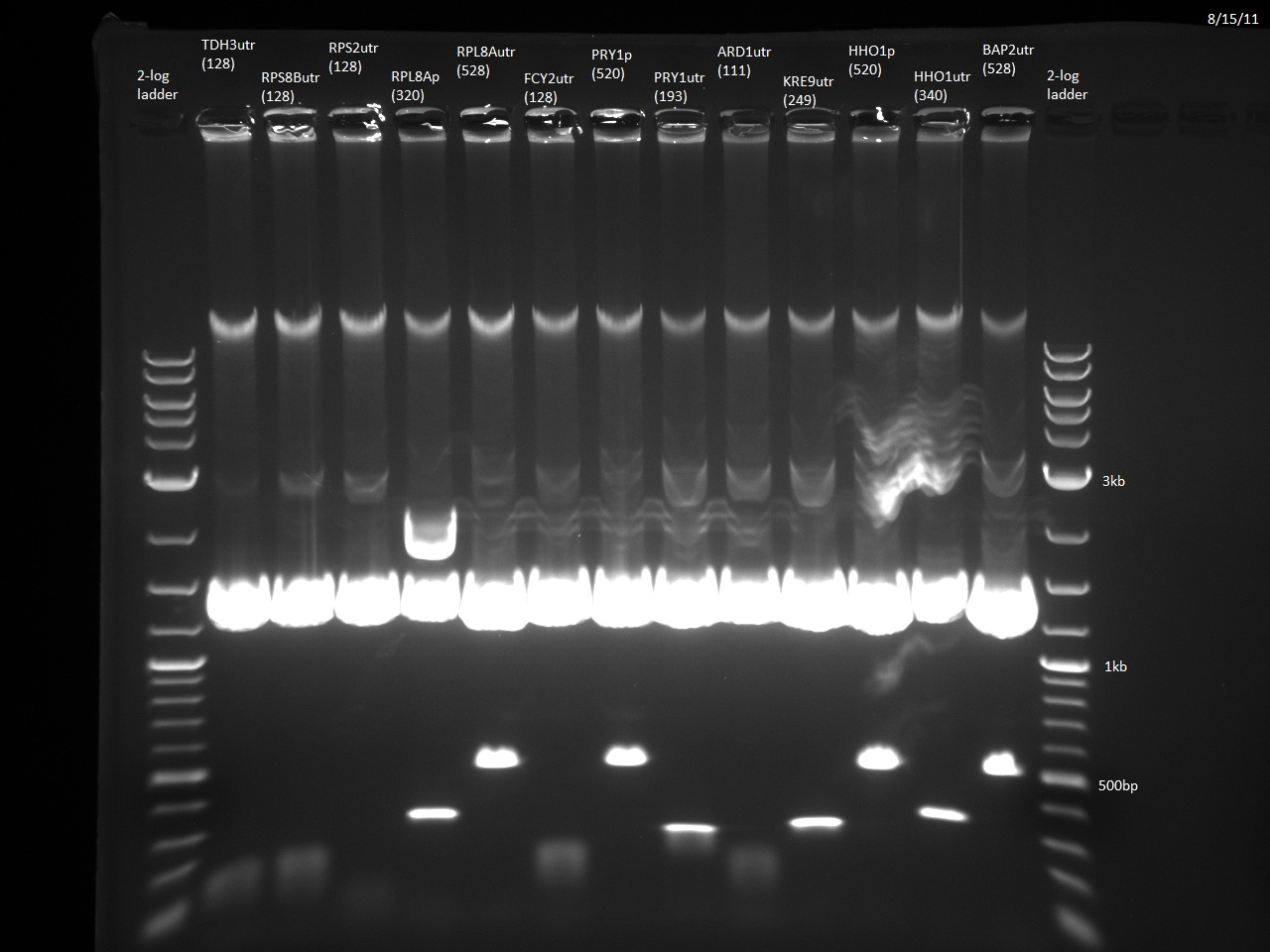
We currently have 23/24 promoters and 3'UTRs from our Golden Gate Assembly-compatible library blunt-end ligated into the vector pTZ19u with SmaI. The promoters and 3'UTRs were PCR'd from yeast genomic DNA with primers containing designed BsaI overhangs. Following miniprep and digestion with BsaI, the promoters and 3'UTRs can be combined with our violacein ORFs. The last fragment, RPS2utr, has had the BsaI overhangs added, and will soon be ligated into the vector and transformed into E. coli. Additionally, the TDH3 3'UTR has been ligated to the URA3 sequence using the NotI site, which will allow the selection of yeast that has taken up our 'expression circles'. Finally, the loxPsym linker has been constructed from two single strand DNA oligos. This means that we are almost ready to do the random ligation to create the combinatorial library of violacein 'expression circles' and integrate them into yeast using Cre-Lox recombination.
 "
"