Team:Washington/Celiacs/Methods
From 2011.igem.org
(→Mutagenizing Kumamolisin) |
(→Kunkel Mutagenesis) |
||
| Line 26: | Line 26: | ||
===Kunkel Mutagenesis=== | ===Kunkel Mutagenesis=== | ||
| - | Using ssDNA as a template, we annealed | + | Using our isolated ssDNA as a template, we annealed our mutagenic oligos in a PCR block set to decrease slowly from the denaturing point of DNA to room temperature to allow for specific binding of our oligos to our template. The oligo harboring the desired point mutations was then extended, using the wild-type gene and associated vector as a template, by T7 DNA polymerase. This hybrid vector, with one strand the native Kumamolisin gene and the other a variant containing point mutations, was transformed into UNG and DUT wild-type E. coli. E. coli with active UNG and DUT has the ability to degrade sections of DNA that contain uracil, and replace them with sections complementary to the opposite strand that contain thymine. Thus, the native Kumamolisin strand that still contained the U’s from the UNG-/DUT- strain was degraded, and the new cells incorporated our desired mutation when synthesizing new DNA from the variant strand. |
| + | |||
| + | By these methods, we were able to construct DNA coding all of our designed variant enzymes. We then grew the cells that carried our vector harboring a given variant, and expressed our engineered enzymes for activity assays. | ||
==Using a Whole Cell Lysate Assay to Test Activity of Mutants== | ==Using a Whole Cell Lysate Assay to Test Activity of Mutants== | ||
Revision as of 21:49, 14 September 2011
Redesigning Kumamolisin to Have Higher Activity at Low pH
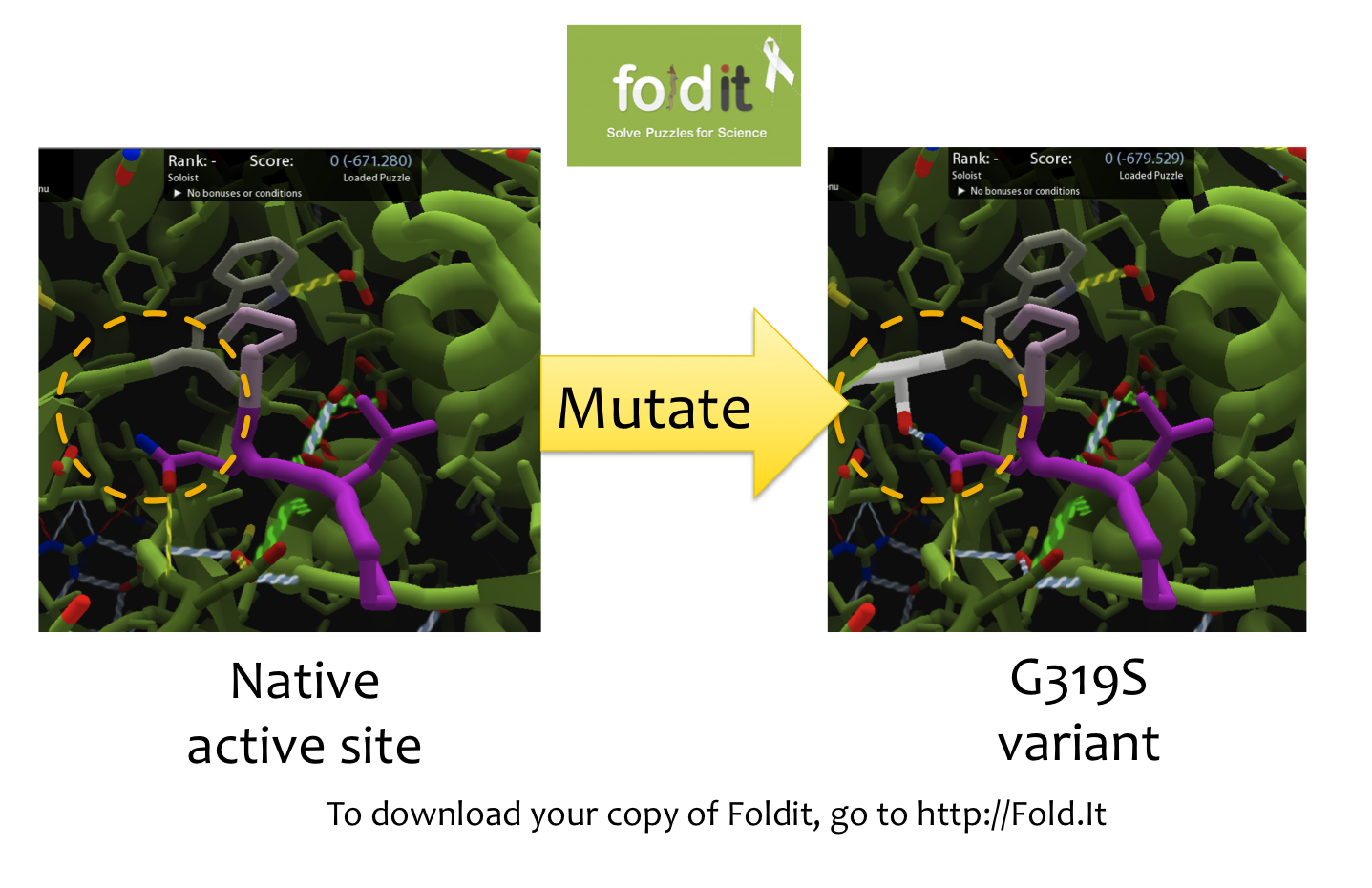
Using Foldit to Design Mutations
In order to design mutations to wild-type Kumamolisin that would increase the enzyme’s proteolytic activity on gluten, we used a computational enzyme editing program called Foldit, which allows the user to hypothetically modify the amino acid sequence of a protein by creating point mutations at any location within the protein’s crystal structure. Within Foldit, we loaded Kumamolisin’s crystal structure in complex with a model PQLP peptide that recurs frequently in gluten, thus mimicking gluten as a substrate. We then modified the amino acid residues around the active site of Kumamolisin in the crystal structure, attempting to decrease the free energy of, and thus stabilize, the system. Estimations of free energy were based on algorithms run by Foldit.
Using this method, we designed over 100 novel mutants, each of which could potentially increase Kumamolisin’s proteolytic activity on gluten.
Mutagenizing Kumamolisin
Kunkel mutagenesis is a classic procedure for incorporating targeted mutations into a piece DNA, so it was ideal for changing our wild-type Kumamolisin gene to code instead for specifically designed variant enzymes.
Producing ssDNA
The first step to producing our specially designed enzymes was to change the wild-type gene that codes for Kumamolisin to code instead for variant enzymes carrying our desired amino acid substitutions. To do this, we designed mutagenic oligonucleotide primers that would anneal to the wild-type Kumamolisin gene and incorporate point mutations that, when expressed, would result in a variant of Kumamolisin with the desired amino acid shift. In order for our primers to anneal to the wild-type gene we isolated the single stranded DNA (ssDNA) of the sense strand of our gene by growing the vector harboring our gene in a uracil-N-glycosidase (UNG) and deoxyuracil triphosphate pyrophosphatase (DUT) deficient strain of E. coli- strain CJ236. Due to the lack of UNG and DUT, these cells to not have the ability to maintain thymine in their DNA, and so all T’s become replaced by U’s in the DNA of these cells.
Once we had the vector harboring our gene grown in UNG-/DUT- cells so that the T’s had been converted to U’s, we harvested the ssDNA of the sense strand by infecting the cells with bacteriophage M13. M13 packages its own 6kb ssDNA genome, identified by length, and so in tandem the phage also packaged our ~6kb vector in single stranded form. We then harvested the phage from the lysed culture of E. coli, and isolated our single stranded vector DNA.
Kunkel Mutagenesis
Using our isolated ssDNA as a template, we annealed our mutagenic oligos in a PCR block set to decrease slowly from the denaturing point of DNA to room temperature to allow for specific binding of our oligos to our template. The oligo harboring the desired point mutations was then extended, using the wild-type gene and associated vector as a template, by T7 DNA polymerase. This hybrid vector, with one strand the native Kumamolisin gene and the other a variant containing point mutations, was transformed into UNG and DUT wild-type E. coli. E. coli with active UNG and DUT has the ability to degrade sections of DNA that contain uracil, and replace them with sections complementary to the opposite strand that contain thymine. Thus, the native Kumamolisin strand that still contained the U’s from the UNG-/DUT- strain was degraded, and the new cells incorporated our desired mutation when synthesizing new DNA from the variant strand.
By these methods, we were able to construct DNA coding all of our designed variant enzymes. We then grew the cells that carried our vector harboring a given variant, and expressed our engineered enzymes for activity assays.
Using a Whole Cell Lysate Assay to Test Activity of Mutants
After the cells had been allowed to grow overnight, colonies were picked and used to inoculate a 96 well plate containing LB and kanamycin. This step allowed us to grow a representative amount of cells containing each mutation. After growing overnight at 37 degrees celcius cells from each well were transferred to another 96 well plate containing TB and kanamycin. These plates were incubated at 37 degrees celcius and later induced using IPTG. After induction, we incubated the plates at 18 degrees celcius overnight. We then lysed the cells and tested the supernatant for proteolytic activity towards PQLP in an assay which measured PQLP degradation over a period of 30 minutes. The assay was done at pH 4 in accordance with the assays done to test ScPEP according to the literature. The mutants were tested against wild-type kumamolisin and ScPEP, an enzyme currently used for the treatment of gluten intolerance via proteolysis. The assay we used was not highly accurate in terms of actual activity. However, what the assay allowed us to do was determine activity relative to our controls. This allowed us to determine which mutants were worth purifying to get more accurate activity data. File:Washington Assay.png
Testing Purified Mutants to Accurately Assess Activity
Purification
After compiling a set of mutants which showed a relative increase in activity we proceeded to purify our mutant proteins. This step is crucial because it allows us to determine how our mutant compares with the wild-type on a quantitative level. For instance, if the whole cell lysate assay showed one mutant to have ten times the activity level as wild-type kumamolisin we cannot assume that there has been an increase in activity because there could simply be ten times the amount of protein with the same level of activity. To purify our mutants, we first grew them in TB and kanamycin with a single colony of the mutant. This inoculation was grown over 24 hours at 37 degrees celcius and then expanded to 50 mililiters (TB+kanamycin). We then allowed the culture to grow to a specific optical density of cells before inducing the culture using IPTG. We then allowed the cells to grow again to express kumamolisin. We then lysed the cells using a lysis buffer and centrifugation. This allowed the enzymes to be released from the cells. The proteins were then collected on a column, washed with buffer, and eluted off the column. Finally, wwe dialyzed the protein in a sodium acetate buffer (pH 4).
Assay
Once we had pure protein, we determined the concentration of each using a NanoDrop machine. With the concentration of each, we then diluted the proteins to the same concentration. For the assay, we also used purified wild-type kumamolisin and ScPEP. The assay was again run for 30 minutes at pH 4. Using the data from the purified assays, we were able to deermine which mutants had higher activity than kumamolisin and by how much their activity was greater.
 "
"