Team:EPF-Lausanne/Our Project/Data
From 2011.igem.org
(→T7 Promoter Variants) |
(→T7 Promoter Variants) |
||
| Line 56: | Line 56: | ||
[http://partsregistry.org/Part:BBa_K613001 K613001] | [http://partsregistry.org/Part:BBa_K613001 K613001] | ||
| + | |||
[http://partsregistry.org/Part:BBa_K613002 K613002] | [http://partsregistry.org/Part:BBa_K613002 K613002] | ||
| + | |||
[http://partsregistry.org/Part:BBa_K613003 K613003] | [http://partsregistry.org/Part:BBa_K613003 K613003] | ||
| + | |||
[http://partsregistry.org/Part:BBa_K613004 K613004] | [http://partsregistry.org/Part:BBa_K613004 K613004] | ||
| + | |||
[http://partsregistry.org/Part:BBa_K613005 K613005] | [http://partsregistry.org/Part:BBa_K613005 K613005] | ||
| + | |||
[http://partsregistry.org/Part:BBa_K613006 K613006] | [http://partsregistry.org/Part:BBa_K613006 K613006] | ||
| + | |||
| + | |||
| + | Variants with additional lac operator (LacI repressed): | ||
| + | |||
| + | [http://partsregistry.org/Part:BBa_K613007 K613007] | ||
| + | |||
| + | [http://partsregistry.org/Part:BBa_K613008 K613008] | ||
| + | |||
| + | [http://partsregistry.org/Part:BBa_K613009 K613009] | ||
| + | |||
| + | [http://partsregistry.org/Part:BBa_K613010 K613010] | ||
| + | |||
| + | [http://partsregistry.org/Part:BBa_K613011 K613011] | ||
| + | |||
| + | [http://partsregistry.org/Part:BBa_K613012 K613012] | ||
=== Characterization of Variants Using Fluorescence === | === Characterization of Variants Using Fluorescence === | ||
Revision as of 13:47, 21 September 2011
Data
team, please check this page: [1]
Contents |
New parts
we must have 3 favourite, then the others come in a "other biobrick" section
Medium-strength Plac
blablabla
TetR Mutants
- V36F
Sequence
ATGTCCAGATTAGATAAAAGTAAAGTGATTAACAGCGCATTAGAGCTGCTTAATGAGGTCGGAATCGAAGGTTTAACAACCCGTAAACTCGCCCAGAAGCTAGGTTTCGAGCAGCCTACATTGTATTGGCATGTAAAAAATAAGCGGGCTTTGCTCGACGCCTTAGCCATTGAGATGTTAGATAGGCACCATACTCACTTTTGCCCTTTAGAAGGGGAAAGCTGGCAAGATTTTTTACGTAATAACGCTAAAAGTTTTAGATGTGCTTTACTAAGTCATCGCGATGGAGCAAAAGTACATTTAGGTACACGGCCTACAGAAAAACAGTATGAAACTCTCGAAAATCAATTAGCCTTTTTATGCCAACAAGGTTTTTCACTAGAGAATGCATTATATGCACTCAGCGCTGTGGGGCATTTTACTTTAGGTTGCGTATTGGAAGATCAAGAGCATCAAGTCGCTAAAGAAGAAAGGGAAACACCTACTACTGATAGTATGCCGCCATTATTACGACAAGCTATCGAATTATTTGATCACCAAGGTGCAGAGCCAGCCTTCTTATTCGGCCTTGAATTGATCATATGCGGATTAGAAAAACAACTTAAATGTGAAAGTGGGTCT
- P39K
- Y42F
- P39Q-Y42M
Sequence
ATGTCCAGATTAGATAAAAGTAAAGTGATTAACAGCGCATTAGAGCTGCTTAATGAGGTCGGAATCGAAGGTTTAACAACCCGTAAACTCGCCCAGAAGCTAGGTGTAGAGCAGCAAACAGTGATGTGGCATGTAAAAAATAAGCGGGCTTTGCTCGACGCCTTAGCCATTGAGATGTTAGATAGGCACCATACTCACTTTTGCCCTTTAGAAGGGGAAAGCTGGCAAGATTTTTTACGTAATAACGCTAAAAGTTTTAGATGTGCTTTACTAAGTCATCGCGATGGAGCAAAAGTACATTTAGGTACACGGCCTACAGAAAAACAGTATGAAACTCTCGAAAATCAATTAGCCTTTTTATGCCAACAAGGTTTTTCACTAGAGAATGCATTATATGCACTCAGCGCTGTGGGGCATTTTACTTTAGGTTGCGTATTGGAAGATCAAGAGCATCAAGTCGCTAAAGAAGAAAGGGAAACACCTACTACTGATAGTATGCCGCCATTATTACGACAAGCTATCGAATTATTTGATCACCAAGGTGCAGAGCCAGCCTTCTTATTCGGCCTTGAATTGATCATATGCGGATTAGAAAAACAACTTAAATGTGAAAGTGGGTCT
T7 promoter variants
Pre-existing parts
add something about the lysis cassette, and add also on its page on the partsregistry!
DNA recovery
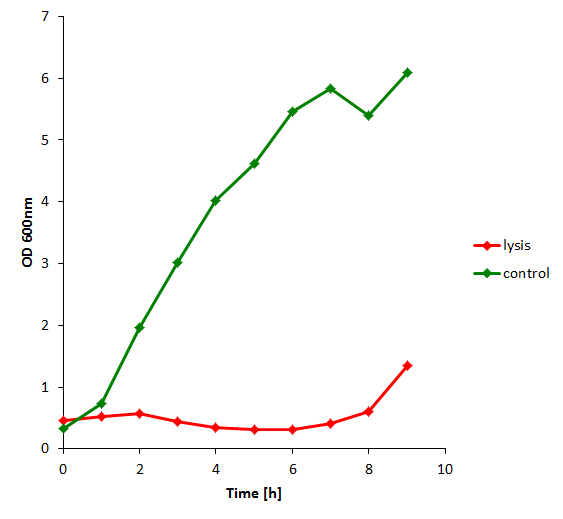
The first experiment for DNA recovery -- lysing cells containing an RFP plasmid, recovering DNA, and counting colonies (or qPCR) -- resulted in confirmation that the lysis approach works for recovering plasmids. Quantitative confirmation of lysing was obtained by taking optical density (OD) measurements every hour using a spectrophotometer. These measurements show that lysing began very shortly after induction with 500 uM IPTG. As expected, the negative control continued to grow unimpeded in the medium.
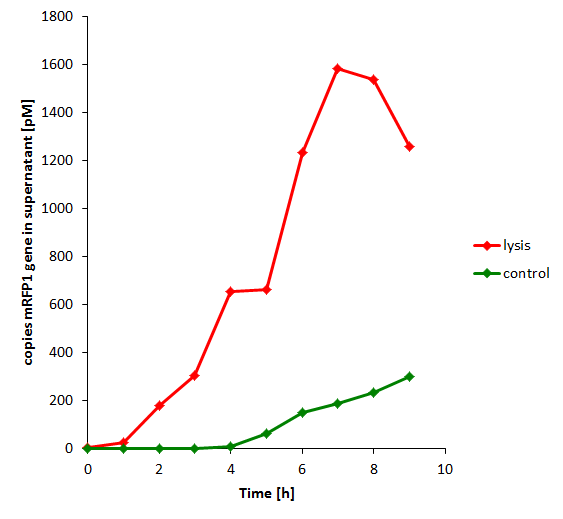
Samples of the broth were collected every hour. These were spun down, and the supernatant was sterile filtered (.22 microns) to clear out all non-DNA contents. These were then used to run a qPCR, with the primers chosen so as to amplify the RFP-containing plasmid that was co-transformed with the lysis-containing plasmid. The results show that the mRFP plasmid was found increasingly in the supernatant as lysing continued.
T7 Promoter Variants
Non-random T7 promoter variants
Variants without additional lac operator (constitutive):
[http://partsregistry.org/Part:BBa_K613001 K613001]
[http://partsregistry.org/Part:BBa_K613002 K613002]
[http://partsregistry.org/Part:BBa_K613003 K613003]
[http://partsregistry.org/Part:BBa_K613004 K613004]
[http://partsregistry.org/Part:BBa_K613005 K613005]
[http://partsregistry.org/Part:BBa_K613006 K613006]
Variants with additional lac operator (LacI repressed):
[http://partsregistry.org/Part:BBa_K613007 K613007]
[http://partsregistry.org/Part:BBa_K613008 K613008]
[http://partsregistry.org/Part:BBa_K613009 K613009]
[http://partsregistry.org/Part:BBa_K613010 K613010]
[http://partsregistry.org/Part:BBa_K613011 K613011]
[http://partsregistry.org/Part:BBa_K613012 K613012]
Characterization of Variants Using Fluorescence
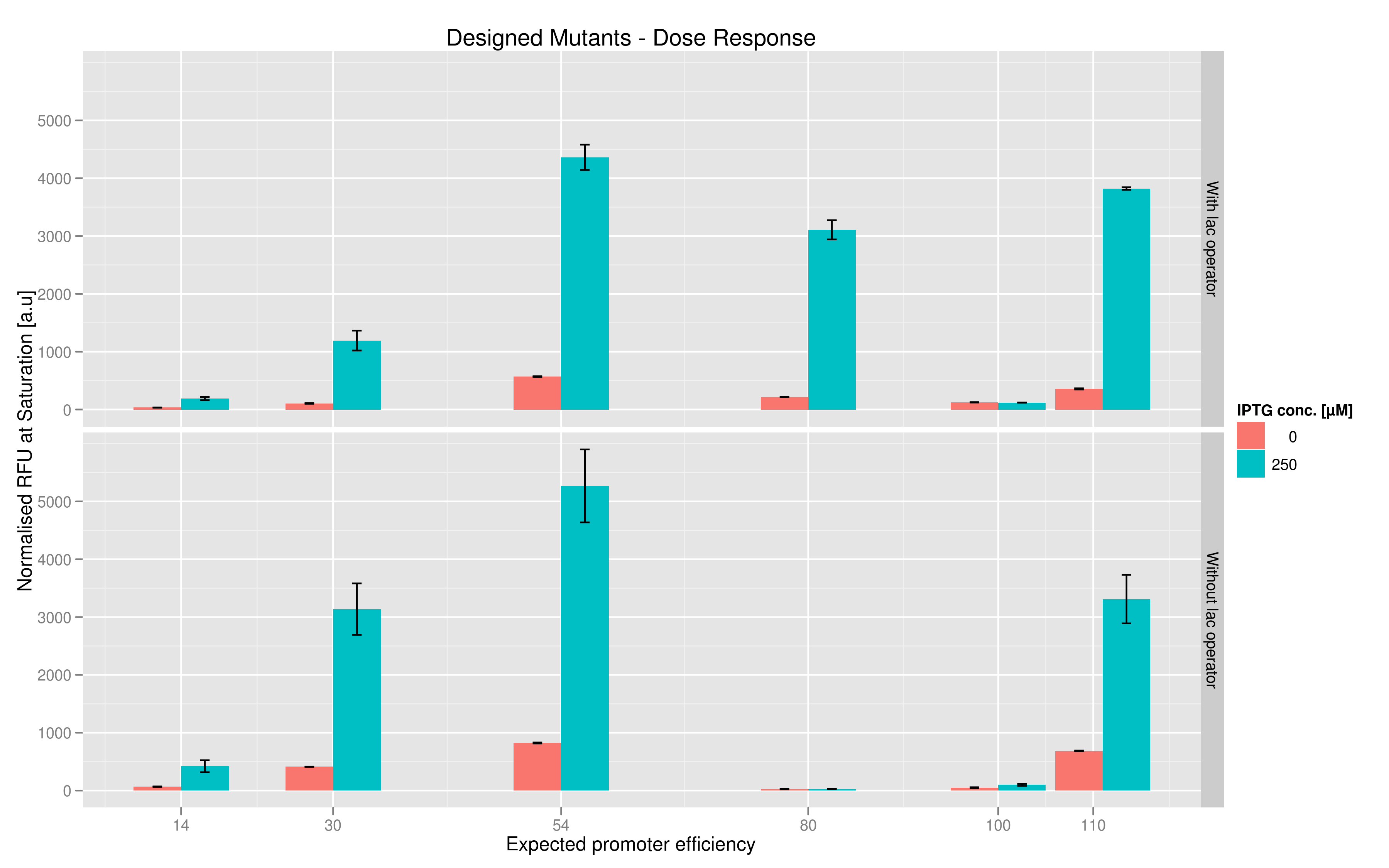
For each family, we tested the randomers and the designed variants separately. To characterize the promoter strengths, we used RFP as the reporter gene and used a platereader to test for fluorescence during and after induction with IPTG.
The six designed T7 promoter variants are named as a function of their predicted promoter efficiency, relative to the wildtype. For example, T7 14 has a predicted efficiency of 14% compared to the consensus T7 promoter, whereas T7 111 is predicted to be 111% more efficient than the wildtype. In the chart above, you find each of the designed promoter variants for both the T7 with and without the lac operator, arranged in increasing predicted efficiency. Contrary to our expectation, some variants that were designed to have a lesser efficiency than the wildtype (e.g. T7 54) seem to have a much higher strength (as measured by fluorescence at saturation, normalized by the optical density). Already in the designed variants, we see a substantial difference in the behaviour of the promoters that have a lac operator as opposed to those that do not. The data for this graph was produced in triplicate, so the error bar represents the standard error across those three measurements.
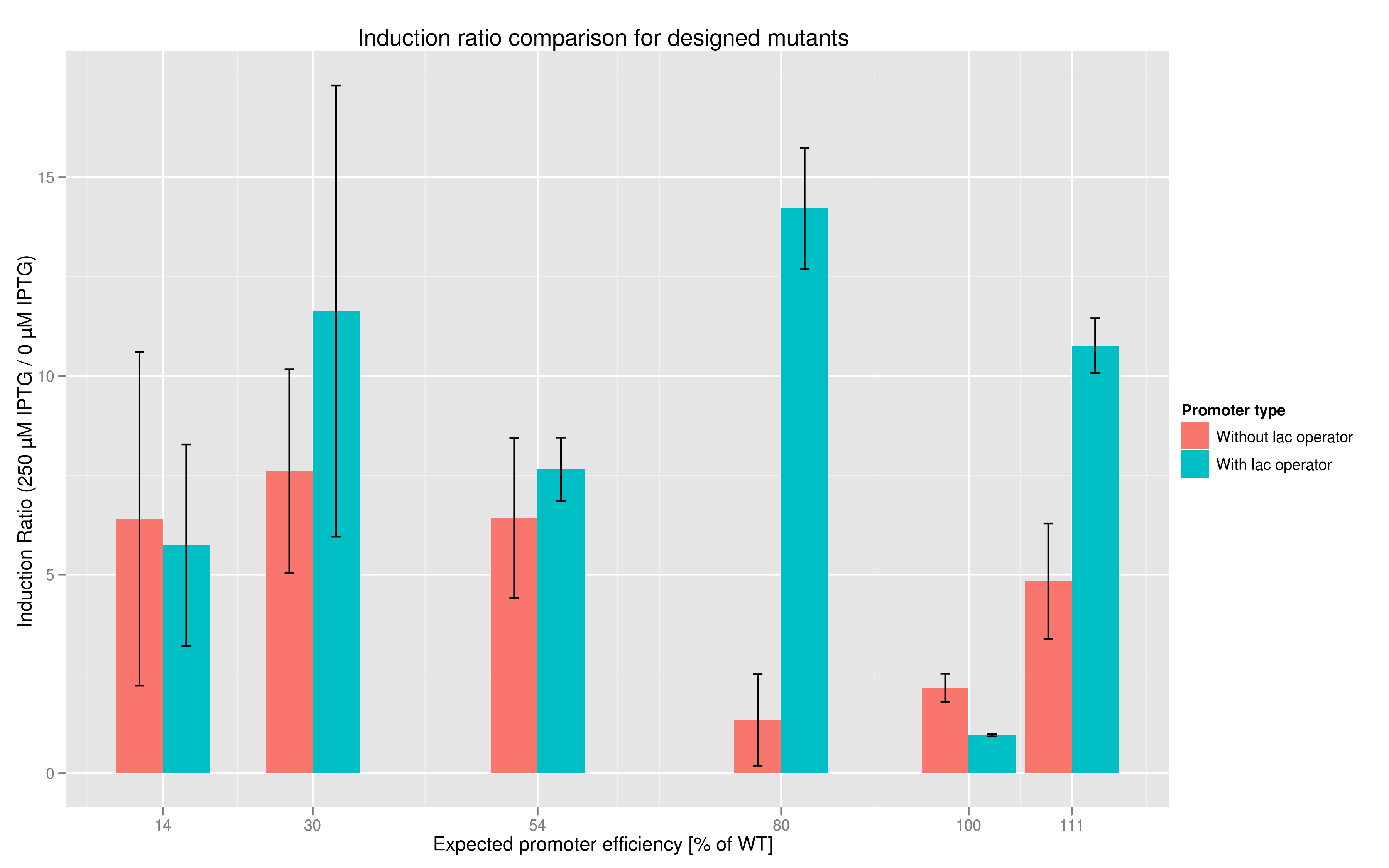
In addition to fluorescence at saturation, another way to characterize promoter strength is to look at its induction ratio, which is the ratio, at saturation, of fluorescence produced by induction with IPTG versus fluorescence produced without induction. In layman's terms, it indicates how strongly the promoter reacts to induction. Here again, our results indicate that some promoter variants (T7 80 in particular) stand out with respect to this feature. Here too the importance of the lac operator in producing high induction ratios is not to be underestimated.
For the three sets of randomers for T7 with and without the lac operator, we tested seventy-two different variants and characterized their expression using the same IPTG induction protocol as with the designed variants. The goal of using these variants was to examine the range of expressions that can be produced by random mutations as opposed to directed mutations. The results, as presented in the graph, indicate that the designed variants (with and without lac operator put together) produce a much higher average normalized fluorescence than the randomers.
Characterization and DNA Recovery with Lysis
Since a major component of our scheme for selecting promoters and transcription factors with strong binding affinities required the ability to lyse cells, we also wanted to test a T7-driven lysis cassette.
Induction with various concentrations of IPTG reveals a steady increase in the amount of lysis that can be obtained. Here 500 uM yields the most substantial amount of lysis, and that concentration was used in all further experiments dealing with lysis. The negative controls were two-fold: one is an unsuccessful attempt at inserting the lysis cassette downstream of a T7 promoter in the psB3K1 plasmid, while the other is a T7 promoter upstream of an RFP gene. Neither should express lysis.
In vivo characterization - Readout systems
We created two different readout systems to characterize TetR mutants in vivo. This is the last step of our whole method, after having selected the interesting TetR-Ptet mutant pairs with the lysis device and having characterized them in vitro.
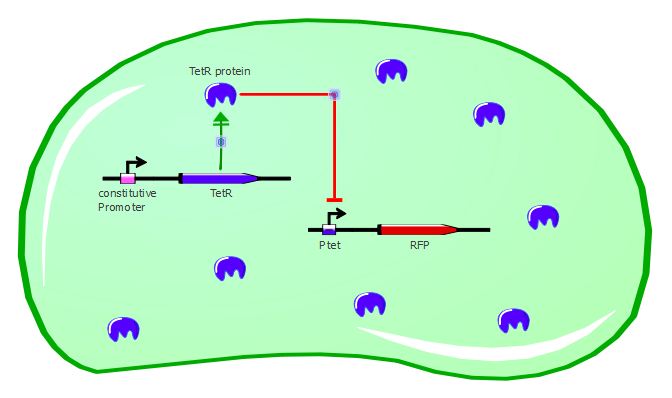
TetR - RFP
The first readout system is composed of TetR with a constitutive promoter, followed by RFP with a Ptet promoter. If TetR binds to Ptet, then RFP is repressed. For more details about them and the experimental results, please refer to the Reporter systems page.
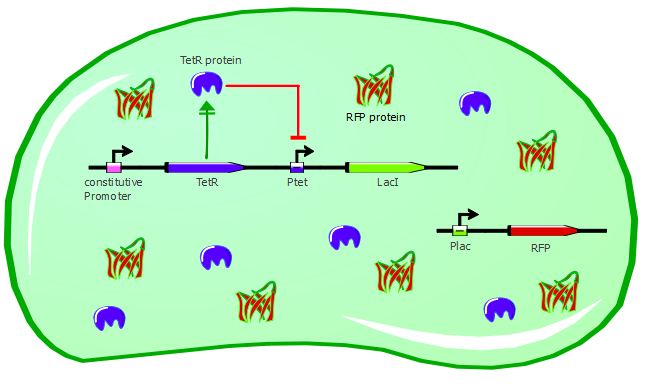
TetR - LacI - RFP
This second readout system is composed of 3 genes: TetR under Pconst control, LacI under Ptet control (playing the role of an inverter) and finally RFP under pLac control. Here, RFP is induced when TetR binds to pTet. The experimental results are available on the Reporter systems page.
 "
"