Team:EPF-Lausanne/Notebook/September2011
From 2011.igem.org
(→Thursday, 8 September 2011) |
|||
| Line 95: | Line 95: | ||
On the other hand, sequencing results for pSB TetR-LacI show that the Ptet promoter before LacI is mutated: we have 3 deletions and one mismatch. The deletions are in the two binding sites of TetR, probably preventing TetR action on the promoter. This can then explain why we had a low RFP expression in the paltereader for the pSB TetR-LacI + J6 Plac-RFP cotransformation: we thought Pconst was too weak, leading to a too big LacI expression, but actually the problem was that TetR couldn't repress well LacI. | On the other hand, sequencing results for pSB TetR-LacI show that the Ptet promoter before LacI is mutated: we have 3 deletions and one mismatch. The deletions are in the two binding sites of TetR, probably preventing TetR action on the promoter. This can then explain why we had a low RFP expression in the paltereader for the pSB TetR-LacI + J6 Plac-RFP cotransformation: we thought Pconst was too weak, leading to a too big LacI expression, but actually the problem was that TetR couldn't repress well LacI. | ||
Nadine made liquid cultures of J61002 Ptet-RFP because we were running out of DNA. We want to send it for sequencing, to be sure that in this plasmid Ptet is OK - although the experiments suggest that yes. She also made liquid cultures of the successful ligations (see yesterday's results) in order to sequence-verify them afterwards. | Nadine made liquid cultures of J61002 Ptet-RFP because we were running out of DNA. We want to send it for sequencing, to be sure that in this plasmid Ptet is OK - although the experiments suggest that yes. She also made liquid cultures of the successful ligations (see yesterday's results) in order to sequence-verify them afterwards. | ||
| + | |||
| + | Vincent transformed T7-const-C2-Lysis and T7-const-C11-Lysis (as a negative control) into BL21 cells and plated them. He also co-transformed: | ||
| + | |||
| + | * T7-const-C2-Lysis + Ptet-GFP | ||
| + | |||
| + | * T7-const-C2-Lysis + Ptet-RFP | ||
| + | |||
| + | * T7-const-C11-Lysis + Ptet-GFP | ||
| + | |||
| + | * T7-const-C11-Lysis + Ptet-RFP | ||
| + | |||
| + | into BL21 using Amp + Kan plates. He used 1 uL of each plasmid. | ||
| + | |||
| + | Vince also prepared a 96 well plate for testing all the randomers (7, 8, 9, 16, 17, 18) of T7-RFP and grew it overnight (shaking, with Easy-Breathe paper to let the cells get air). | ||
== Friday, 09 september 2011 == | == Friday, 09 september 2011 == | ||
Revision as of 11:36, 12 September 2011
Notebook: September 2011
Contents |
Thursday, September 1 2011
Vincent PCR purified the pSB3K1-extended (no TetR) for use in the Gibsons and negative controls.
The Gibson assembly of J6-Plac-Lysis had produced a single colony. Vincent colony-PCRed it using two different sets of primers to amplify subportions of the lysis cassette. The first set of primers (30-f & 816-r) amplify an 800 bp piece whereas the second set (Seq-J6-RFP-f & Seq-J6-RFPlysis-r) amplifies a 1100 bp piece.
The gel seems to confirm that lysis is in the plasmid. We will send the plasmid for sequencing on Friday.
After taking a closer look at the plate from the platereader, it seems that none of the T7 variants did any lysing. This realization made it urgent that we discover whether or not the full T4-lysis cassette was really in these Gibson-assembled plasmids. The samples that had been sent in for sequencing had primers that seemed to amplify the backbone, instead of the lysis cassette. While this does not necessarily mean that the lysis is not there, it does suggest that all the PCRS and gels used to verify the presence of lysis were only showing the existence of the backbone.
Alina ran a PCR with different primers from Douglas' first attempts at putting together the 2700 bp cassette that amplify select regions of the cassette.
Friday, September 2 2011
Alina wanted to try a new extension PCR of the T4-lysis and a Gibson of the exteneded-lysis into the K1 backbone. The transformation was successful with tons of colonies. A colony PCR will be run on Monday.
Nadine came back in the lab and cotransformed pSB3K1 Pconst-TetR with J61002 Plac-RFP since the sequencing results were concluent, in order to perform IPTG and ATC platereader experiments.
We had a lot of contaminated bottles, so she also made new SOC bottles.
Monday, September 5 2011
Alina ran a colony PCR on the K1-T7-lysis Gibson assembly.
After having set liquid cultures on Saturday, Nadine set up the platereader experiment as follows:
- J61002 Plac-RFP with increasing concentrations of IPTG
- pSB3K1 TetR-LacI + J61002 Plac-RFP with increasing concentrations of IPTG
- pSB3K1 TetR-LacI + J61002 Plac-RFP with increasing concentrations of ATC
She had also prepared co-transformations with pSB3K1 TetR-LacI + J61002 Plac-lysis, but the sequencing results indicated that the lysis cassette was wrong.
Tuesday, 6 September 2011
The results from the platereader experiment came back and they look good!
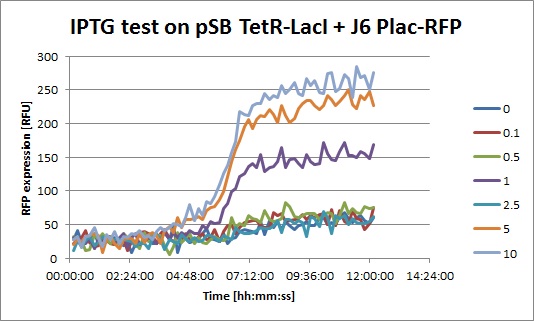
- J6 Plac-RFP: The level of RFP expression doesn't rise much with high doses of IPTG, showing that DH5alpha cells don't express much LacI and that Plac is a quite weak promoter.
- J6 Plac-RFP + pSB TetR-LacI, IPTG: you can clearly see an increase of RFP when IPTG is added. This means that even with a constitutive promoter in front of TetR, LacI is still expressed. In fact, Pconst is weaker than Ptet. The "2.5" data seems strange, I perhaps missed to put IPTG in this well.
- J6 Plac-RFP + pSB TetR-LacI, ATC: Without ATC, we have a similar expression curve as in the IPTG experiment. When ATC is added, we see a diminution of RFP expression because LacI is not repressed anymore.
Vincent transformed the "randomer" Gibson assemblies (from nearly a month ago) into BL21 cells. If you cross-check with the numerical scheme, those would be variants 7, 8, 9, 16, 17, 18.
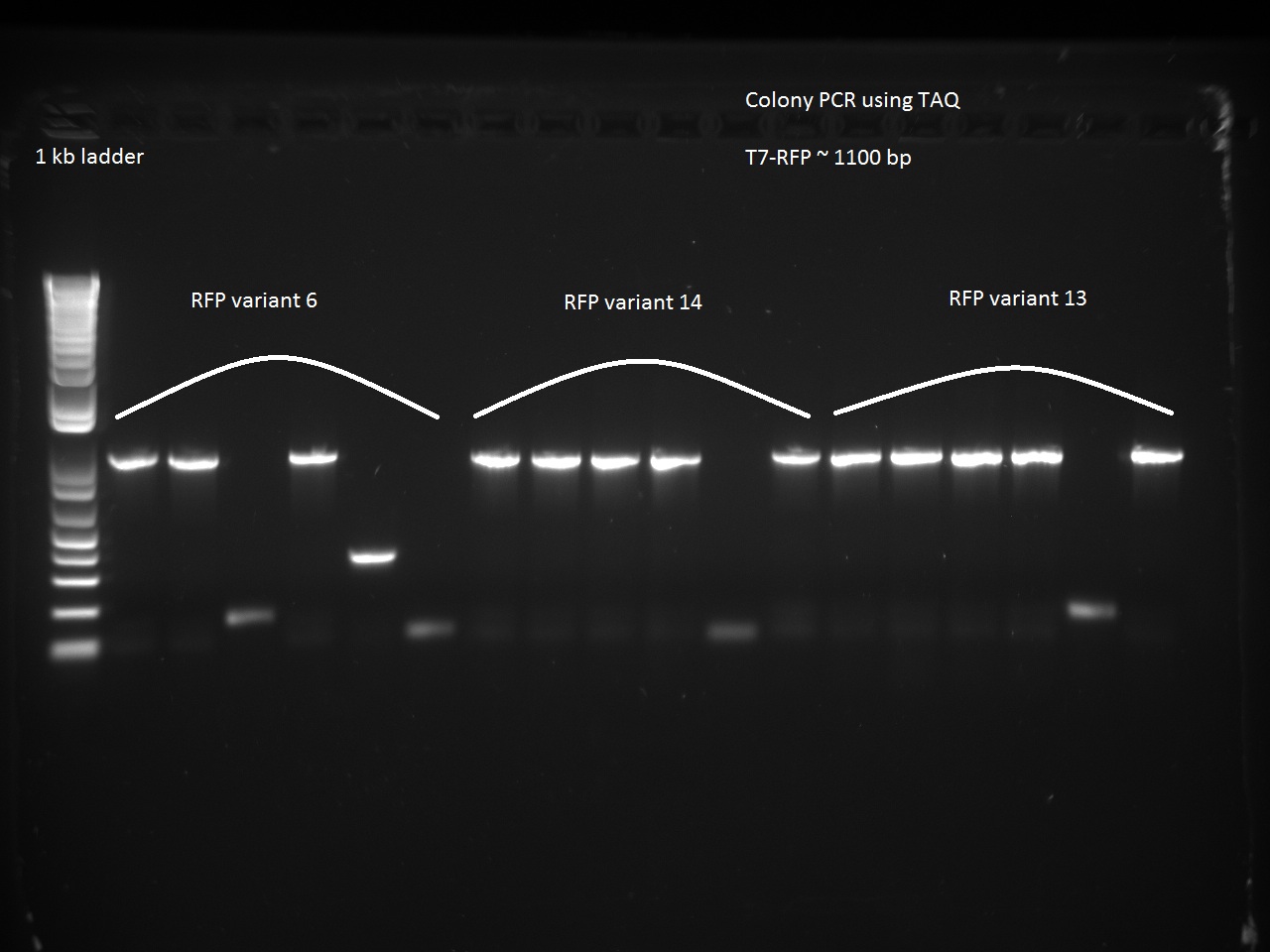
We also went after the remaining non-randomer variants that were left (6, 13, and 14) using a colony PCR on six colonies of each variant. The PCR used 64 C for annealing and 1 minute for extension.
Wednesday, 7 September 2011
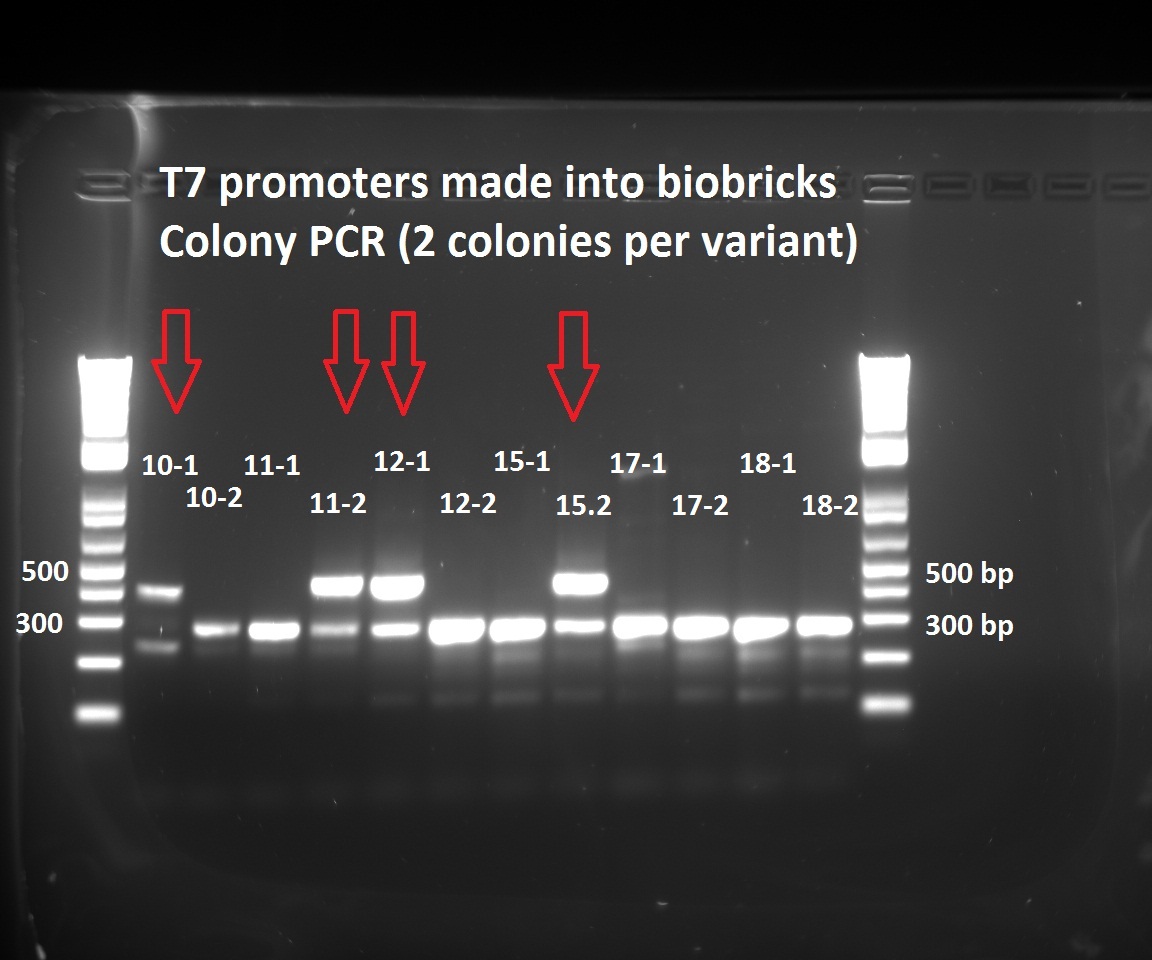
Nadine made colony PCRs for the T7 promoter variants that Henrike cloned into the biobrick vector. The right insert should be ~450 bp long, but all the colonies show a fragment at 300 bp. Nevertheless, we have 5 colonies with the 450 insert. The low number of positive colonies correlates with the observation that the ligation negative control plate had about half as much colonies as the different ligations.
She also prepared some liquid cultures for tomorrow's platereader experiment, and miniprepped the pSB TetR-LacI plasmid before sending it for 2 new sequencing reactions.
Vdog made glycerol stocks and minipreps of the non-randomer RFP variants 6, 13, and 14 as well as Alina's T7-const-C2/C11, T7 lac 1, and T7 lac2 3.
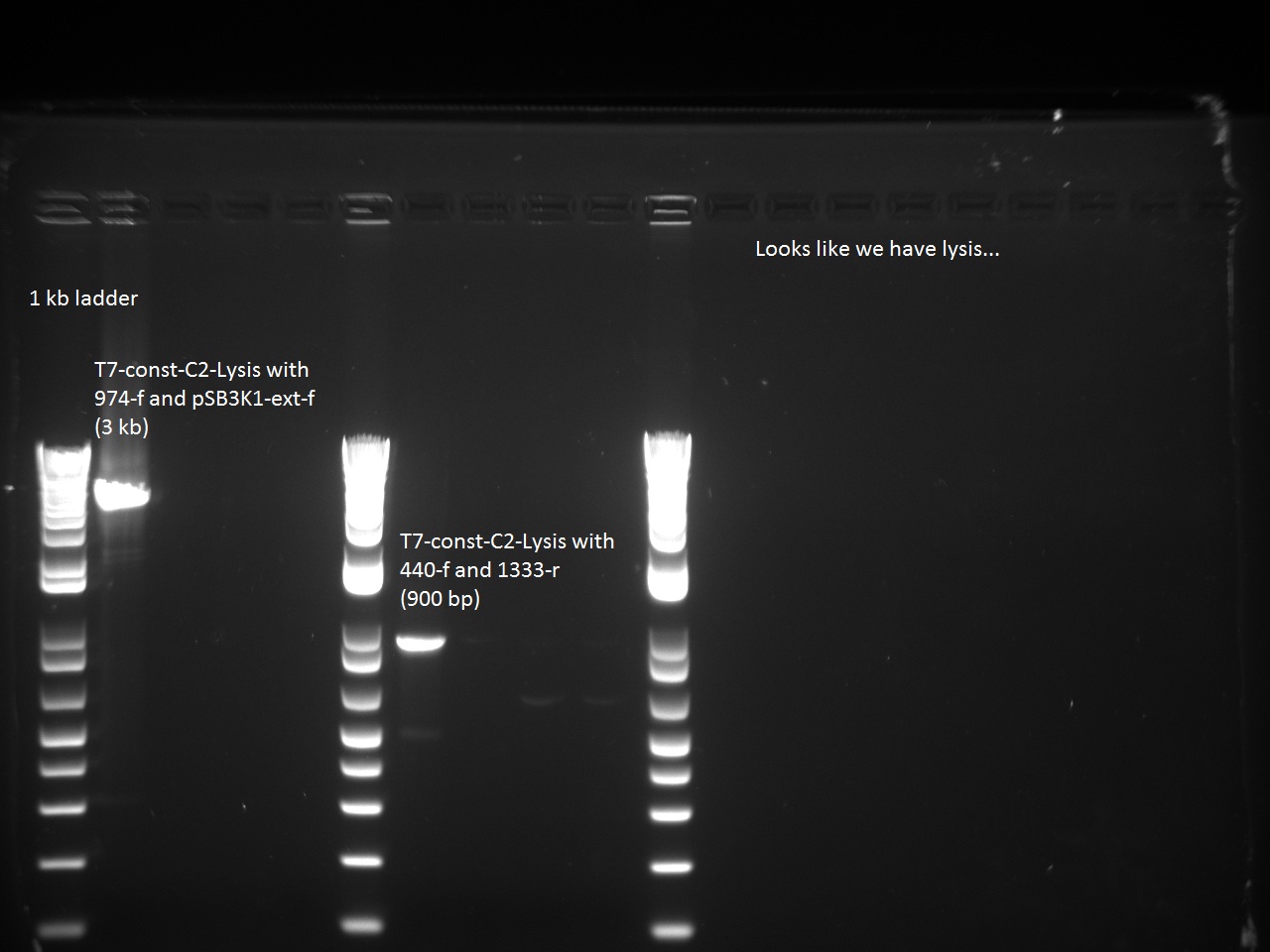
He also sent all of Alina's constructs (aforementioned) for sequencing using two different primers: 947_f and 816_r. He then ran a control PCR on all the T7 constructs using Alina's EPFL TAQ with the following combination of primers:
- 440-f and 1333-r
- 947-f and pSB3K1-ext-r
The protocol asked for a 25 uL aliquot with
- 2.5 uL Thermo Pol buffer (TPB)
- 0.5 uL dNTP
- 0.5 uL primer 1
- 0.5 uL primer 2
- 1 uL plasmid
- 0.25 uL TAQ
He went on to transform the non-randomer RFP minipreps and the T7-const-c2/c11, lac1, and lac2 3 constructs into BL21 cells This was followed by the production of a 96 round-bottom well plate with 100 uL of LB with 1 uL of Kan. Twelve colonies from each plate were chosen and swirled into the individual wells. This plate was put on a shaker overnight (after having been sealed with the Easy-Breathe adhesive paper).
Thursday, 8 September 2011
Sequencing of T7-const-C2 returned the correct sequence, as did the Control PCR, so it seems that the lysis cassette has found its way into the T7-const-C2!
On the other hand, sequencing results for pSB TetR-LacI show that the Ptet promoter before LacI is mutated: we have 3 deletions and one mismatch. The deletions are in the two binding sites of TetR, probably preventing TetR action on the promoter. This can then explain why we had a low RFP expression in the paltereader for the pSB TetR-LacI + J6 Plac-RFP cotransformation: we thought Pconst was too weak, leading to a too big LacI expression, but actually the problem was that TetR couldn't repress well LacI.
Nadine made liquid cultures of J61002 Ptet-RFP because we were running out of DNA. We want to send it for sequencing, to be sure that in this plasmid Ptet is OK - although the experiments suggest that yes. She also made liquid cultures of the successful ligations (see yesterday's results) in order to sequence-verify them afterwards.
Vincent transformed T7-const-C2-Lysis and T7-const-C11-Lysis (as a negative control) into BL21 cells and plated them. He also co-transformed:
- T7-const-C2-Lysis + Ptet-GFP
- T7-const-C2-Lysis + Ptet-RFP
- T7-const-C11-Lysis + Ptet-GFP
- T7-const-C11-Lysis + Ptet-RFP
into BL21 using Amp + Kan plates. He used 1 uL of each plasmid.
Vince also prepared a 96 well plate for testing all the randomers (7, 8, 9, 16, 17, 18) of T7-RFP and grew it overnight (shaking, with Easy-Breathe paper to let the cells get air).
Friday, 09 september 2011
Nadine's platereader experiment gave disappointing results: the cells didn't grow much, reaching only 0.1-0.2 OD instead of 1 normally. This will be repeated next Tuesday. The minipreps of J6 Ptet-RFP and the T7 biobricks yielded high concentrations, and therefore were sent for sequencing.
 "
"