|
|
| (36 intermediate revisions not shown) |
| Line 24: |
Line 24: |
| | | | |
| | <gallery widths=156 perrow=3> | | <gallery widths=156 perrow=3> |
| - | File:Dough_media-negative_control.jpg | + | File:Dough_media-negative_control.jpg|Negative control pure agar |
| - | File:Dough_media_1.jpg | + | File:Dough_media_1.jpg|0.25g flour in plate |
| - | File:Dough_media_2.jpg | + | File:Dough_media_2.jpg|0.35g flour in plate |
| - | File:Dough_media_3.jpg | + | File:Dough_media_3.jpg|0.45g flour in plate |
| - | File:Dough_media_4.jpg | + | File:Dough_media_4.jpg|0.55g flour in plate |
| - | File:Dough media-pos ctrl.jpg | + | File:Dough media-pos ctrl.jpg|Positive control YPD |
| | </gallery> | | </gallery> |
| | | | |
| | | | |
| | [[File:IMG 8910 2.jpg|thumb|200px|Bread baked from Vitamin A yeast.]] | | [[File:IMG 8910 2.jpg|thumb|200px|Bread baked from Vitamin A yeast.]] |
| - | On a first visual comparison of growth on dough media vs YPD we did see a significantly slower growth on dough media than on YPD. This is to be expected considering there is a large excess of nutrients on the YPD plates. There was still significant growth on the dough media plates however, enough to show that it would be viable to use in bread. | + | On a first visual comparison of growth on dough media vs YPD (standard yeast media) we did see a significantly slower growth on dough media than on YPD. This is to be expected considering there is a large excess of nutrients on the YPD plates. There was still significant growth on the dough media plates however, enough to show that it would be viable to use in bread. |
| | | | |
| | We then took it one step further and showed that baking bread with the Vitamin A yeast was possible. | | We then took it one step further and showed that baking bread with the Vitamin A yeast was possible. |
| Line 40: |
Line 40: |
| | =====Spectroscopy and HPLC===== | | =====Spectroscopy and HPLC===== |
| | | | |
| - | [[File:Beta carotene time2.jpg|300px|thumb|left|Beta carotene vs. Time for yeast strains 39, 128, 179, 201[1]]]
| + | We performed spectroscopy experiments, where we took measurements of the number of cells and amount of beta carotene produced at different time points. Beta carotene was extracted using hexane, which only isolates highly hydrophobic molecules like carotenoids. We performed our experiments on 4 beta carotene producing strains with different genes inserted and also a wild type strain. Based on the results, we can see that the production of beta-carotene over time increases at a linear rate, as the number of cells producing beta carotene increases. Production levels off at the later time points, corresponding to the observation that the media gets saturated. All genetically modified strains showed significantly more beta carotene production than the wild type strain. |
| | | | |
| - | [[File:Beta carotene time.jpg|300px|thumb|left|Beta carotene/cell vs. Time for yeast strains 39, 128, 179, 201[1]]] | + | [[File:Beta carotene time2.jpg|600px|thumb|left|Beta carotene vs. Time for yeast strains 39, 128, 179, 201[1]]] |
| | | | |
| - | [[File:HPLC.jpg|300px|thumb|left|Beta carotene extracted from yeast strain 210]]
| + | The normalized beta carotene/cell curve shows a fairly constant amount of beta carotene in each cell. It also provides initial conditions for our model used to optimize the beta carotene pathway. |
| | | | |
| - | Based on the results of our Spectroscopy experiments measuring beta-carotene versus time and characterization by the use of HPLC, we can see that the production of beta-carotene over time increases at a linear rate. The results of the HPLC confirm that the yeast cells are in fact producing beta carotene, as the absorption peak at 450nm corresponds to the absorption maxima of beta-carotene. Before running the samples on the HPLC, beta carotene was extracted from cell extract using hexane, as it would only isolate highly hydrophobic molecules, such as beta-carotene. As seen in following Spectroscopy and HPLC graphs, beta carotene production and characterization can be quantified. In addition, beta carotene per cell over time can be calculated, which provides initial conditions for our model used to optimize the beta carotene pathway. As seen in the HPLC absorbence spectra on the left, beta carotene shows a strong signal at 450nm, matching beta carotene characterization in literature, allowing us to confirm that the orange color produced in our strains can be attributed to increased beta carotene production.
| + | [[File:Beta carotene time.jpg|600px|thumb|left|Beta carotene/cell vs. Time for yeast strains 39, 128, 179, 201[1]]] |
| | | | |
| - | =Protein Purification=
| + | We verified the presence of beta carotene in our extracts by performing HPLC analysis. A reverse phase column was used, as it provides great sensitivity and accuracy, allowing us to isolate beta carotene from other similar carotenoids that may be present. Beta-carotene elutes at around 16.6 minutes, and our sample peaks were compared to those of the standards. HPLC also allows us to quantify beta-carotene accurately. Results from the HPLC analysis agreed with the spectroscopy results. A possible lycopene peak was also detected in the extracts from strain 39. |
| | | | |
| - | [[File:1st PCR R1 edited.jpg|300px|thumb|left|1st round PCR Gel. Lanes from left to right: 2kb ladder, BTS1, crtE, crtI, crtYB]]
| |
| | | | |
| - | In order to perform enzyme kinetics assays, we needed to first purify our proteins of interest. From the beta carotene pathway, these enzymes were geranylgeranyl pyrophosphate synthase, phytoene desaturase, and a bifunctional phytoene synthase. Using a TEV-his6X-mCherry-KAN tag, we would be able to purify our enzyme with only one high efficiency purification step (Nickel Column Purification using His-tag) and characterize product with great accuracy through the use of anti-mCherry antibodies on the RFP component of the tag. The goal of the PCR was to first produce a PCR product that contained the HIS-TEV-mCherry-KAN-CYC1tt tag. Another PCR would enable us to tag each of our genes of interest, which we could then transform into yeast for expression and purification. As depicted in the two gels to the left, both rounds of PCR proved to be successful, enabling us to transform these tagged genes into yeast.
| + | [[File:Standard 2.png|thumb|400px|center|Beta carotene standard, 70ug/ml]] |
| | | | |
| | + | [[File:Sample 5 in succession.png|thumb|400px|center|Strain 201 extracts. From top: extracted at 19 hours, 44 hours, 65.5 hours, 92 hours,112.5 hours]] |
| | | | |
| - | [[File:1st PCR R2.jpg|300px|thumb|right|2nd round PCR Gel: Lanes from left to right: 2kb ladder, BTS1, crtE, crtI, crtYB]] | + | [[File:Sample 2 in succession.png|thumb|400px|center|Strain 39 extracts. From top: extracted at 19 hours, 44 hours, 65.5 hours, 112.5 hours,115 hours]] |
| | | | |
| - | [[File:Microscopy Images.jpg|400px|thumb|left|Microscopy Images from tagged crtYB-mCherry tagged yeast and control]]
| + | =====Protein Purification===== |
| | | | |
| | + | [[File:1st PCR R1 edited.jpg|180px|thumb|left|1st round PCR Gel. Lanes from left to right: 2kb ladder, BTS1, crtE, crtI, crtYB]] |
| | | | |
| | + | In order to perform enzyme kinetics assays, we needed to first purify our proteins of interest. From the beta carotene pathway, these enzymes were geranylgeranyl pyrophosphate synthase, phytoene desaturase, and a bifunctional phytoene synthase. Using a TEV-his6X-mCherry-KAN tag, we would be able to purify our enzyme with only one high efficiency purification step (Nickel Column Purification using His-tag) and characterize the product with great accuracy through the use of anti-mCherry antibodies on the mCherry component of the tag. The goal of the PCR was to first produce a PCR product that contained the HIS-TEV-mCherry-KAN-CYC1tt tag. Another PCR would enable us to tag each of our genes of interest, which we could then transform into yeast for expression and purification. As depicted in the two PCR gels, both rounds of PCR proved to be successful, as we obtained a large signal for each of our tagged genes. These results enabled us to transform these tagged genes into yeast. |
| | | | |
| - | =References= | + | [[File:1st PCR R2.jpg|180px|thumb|right|2nd round PCR Gel: Lanes from left to right: 2kb ladder, BTS1, crtE, crtI, crtYB]] |
| | + | |
| | + | After transformation of our tagged genes, fluorescence microscopy was used to confirm the success of the transformation and expression of our tagged genes. Depicted in the fluorescence microscopy figure, the transformed yeast cells fluoresce red, which is evidence that our tagged mCherry (RFP) tagged genes are in fact present and being expressed in our yeast. Green fluorescence is seen in both wild type and transformed yeast, which is due to the autofluorescence inherent to beta carotene. In addition, the DIC (differential interference contrast) microscopy image shows the localization of our highly hydrophobic beta carotene molecules inside lipid droplets of the yeast cell. |
| | + | |
| | + | |
| | + | [[File:Microscopy Images.jpg|380px|thumb|center|Microscopy Images from tagged crtYB-mCherry tagged yeast and crtYB control]] |
| | + | |
| | + | =====References===== |
| | | | |
| | 1. High-Level Production of Beta-Carotene in Saccharomyces cerevisiae by Successive Transformation with Carotenogenic Genes from Xanthophyllomyces dendrorhous | | 1. High-Level Production of Beta-Carotene in Saccharomyces cerevisiae by Successive Transformation with Carotenogenic Genes from Xanthophyllomyces dendrorhous |
| | René Verwaal,1, Jing Wang,1 Jean-Paul Meijnen,1 Hans Visser,1,Gerhard Sandmann,2 Johan A. van den Berg,1, and Albert J. J. van Ooyen1* | | René Verwaal,1, Jing Wang,1 Jean-Paul Meijnen,1 Hans Visser,1,Gerhard Sandmann,2 Johan A. van den Berg,1, and Albert J. J. van Ooyen1* |
| | + | |
| | | | |
| | <html> | | <html> |
| | </div> | | </div> |
Results
The strains transformed with the caretenogenic genes are a bright orange color due to all the beta-carotene they produce, as beta-carotene is an orange pigment. We confirmed its presence via HPLC using a standard.
Negative control pure agar
|
|
|
|
|
|

Bread baked from Vitamin A yeast.
On a first visual comparison of growth on dough media vs YPD (standard yeast media) we did see a significantly slower growth on dough media than on YPD. This is to be expected considering there is a large excess of nutrients on the YPD plates. There was still significant growth on the dough media plates however, enough to show that it would be viable to use in bread.
We then took it one step further and showed that baking bread with the Vitamin A yeast was possible.
Spectroscopy and HPLC
We performed spectroscopy experiments, where we took measurements of the number of cells and amount of beta carotene produced at different time points. Beta carotene was extracted using hexane, which only isolates highly hydrophobic molecules like carotenoids. We performed our experiments on 4 beta carotene producing strains with different genes inserted and also a wild type strain. Based on the results, we can see that the production of beta-carotene over time increases at a linear rate, as the number of cells producing beta carotene increases. Production levels off at the later time points, corresponding to the observation that the media gets saturated. All genetically modified strains showed significantly more beta carotene production than the wild type strain.
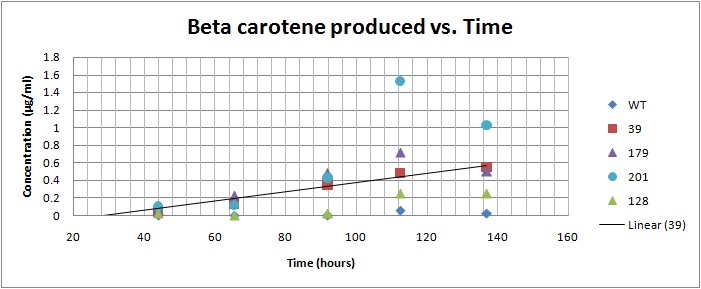
Beta carotene vs. Time for yeast strains 39, 128, 179, 201[1]
The normalized beta carotene/cell curve shows a fairly constant amount of beta carotene in each cell. It also provides initial conditions for our model used to optimize the beta carotene pathway.
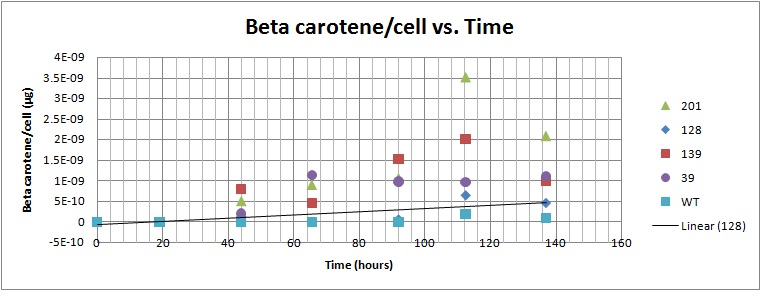
Beta carotene/cell vs. Time for yeast strains 39, 128, 179, 201[1]
We verified the presence of beta carotene in our extracts by performing HPLC analysis. A reverse phase column was used, as it provides great sensitivity and accuracy, allowing us to isolate beta carotene from other similar carotenoids that may be present. Beta-carotene elutes at around 16.6 minutes, and our sample peaks were compared to those of the standards. HPLC also allows us to quantify beta-carotene accurately. Results from the HPLC analysis agreed with the spectroscopy results. A possible lycopene peak was also detected in the extracts from strain 39.
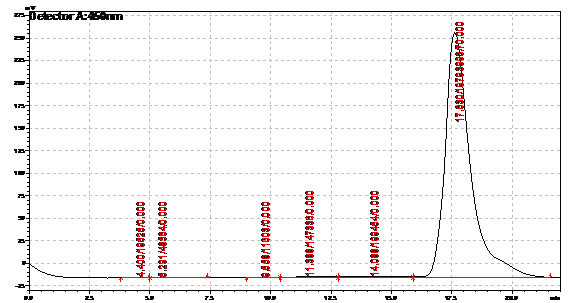
Beta carotene standard, 70ug/ml
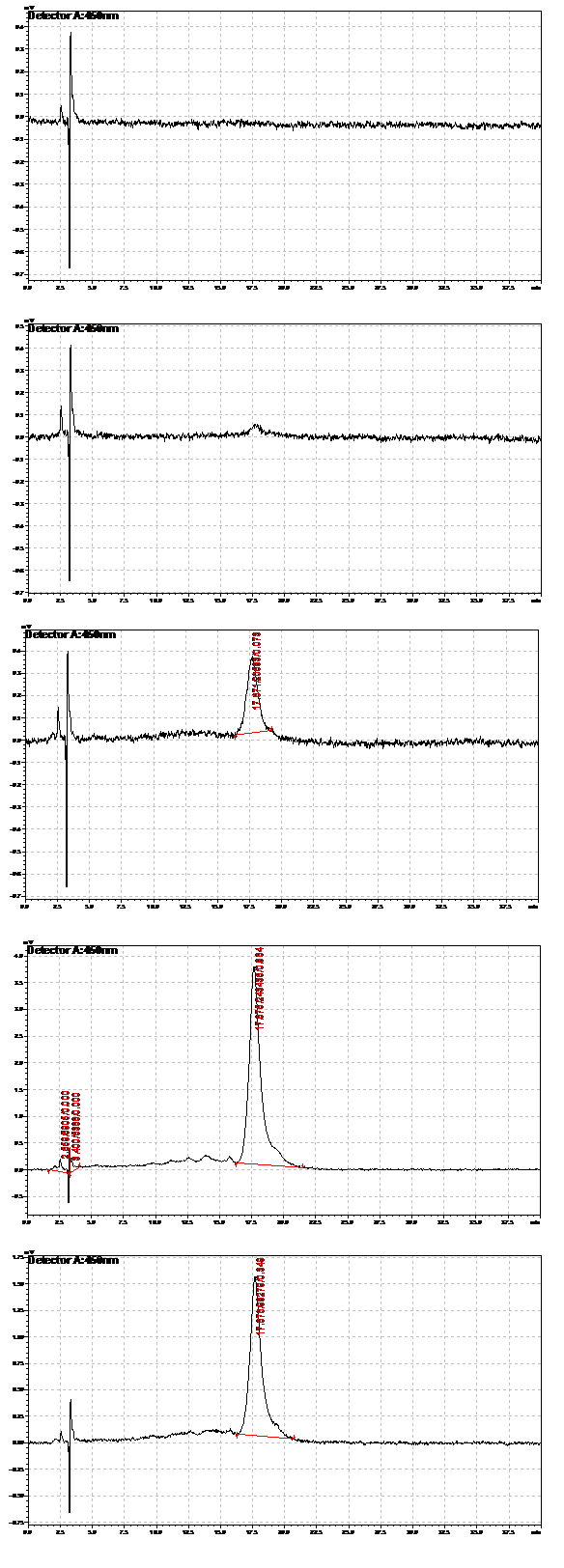
Strain 201 extracts. From top: extracted at 19 hours, 44 hours, 65.5 hours, 92 hours,112.5 hours
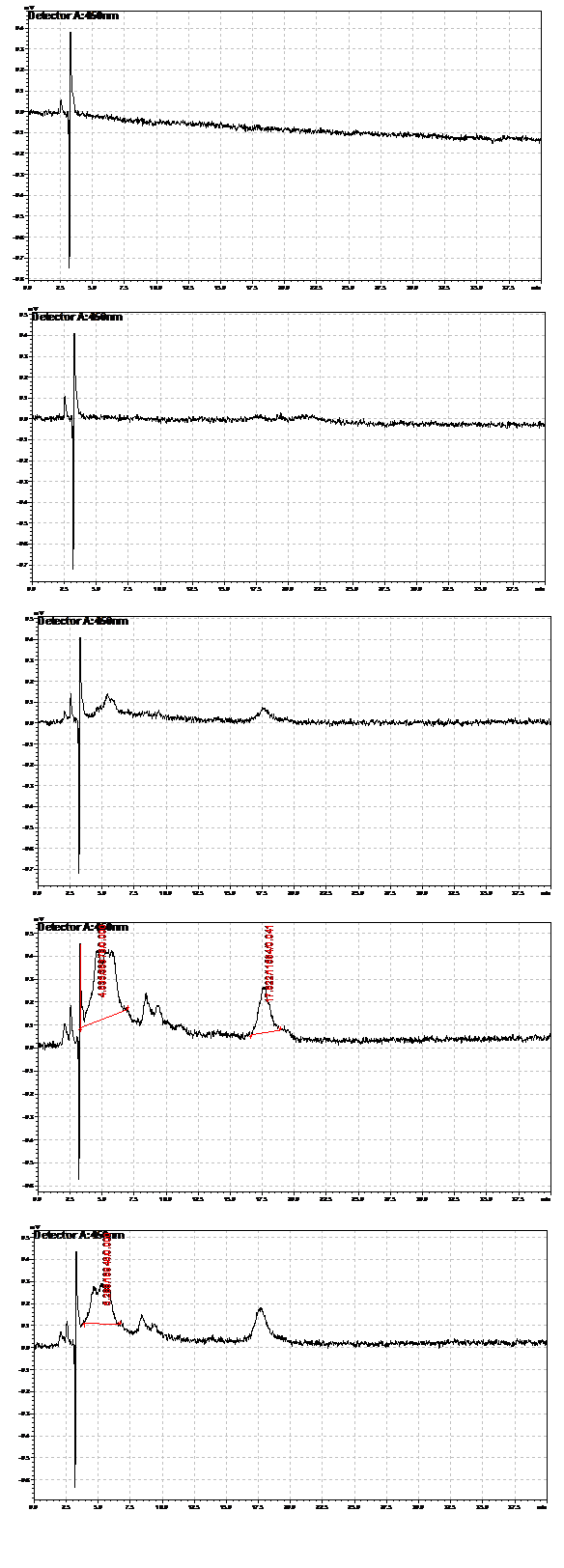
Strain 39 extracts. From top: extracted at 19 hours, 44 hours, 65.5 hours, 112.5 hours,115 hours
Protein Purification
1st round PCR Gel. Lanes from left to right: 2kb ladder, BTS1, crtE, crtI, crtYB
In order to perform enzyme kinetics assays, we needed to first purify our proteins of interest. From the beta carotene pathway, these enzymes were geranylgeranyl pyrophosphate synthase, phytoene desaturase, and a bifunctional phytoene synthase. Using a TEV-his6X-mCherry-KAN tag, we would be able to purify our enzyme with only one high efficiency purification step (Nickel Column Purification using His-tag) and characterize the product with great accuracy through the use of anti-mCherry antibodies on the mCherry component of the tag. The goal of the PCR was to first produce a PCR product that contained the HIS-TEV-mCherry-KAN-CYC1tt tag. Another PCR would enable us to tag each of our genes of interest, which we could then transform into yeast for expression and purification. As depicted in the two PCR gels, both rounds of PCR proved to be successful, as we obtained a large signal for each of our tagged genes. These results enabled us to transform these tagged genes into yeast.
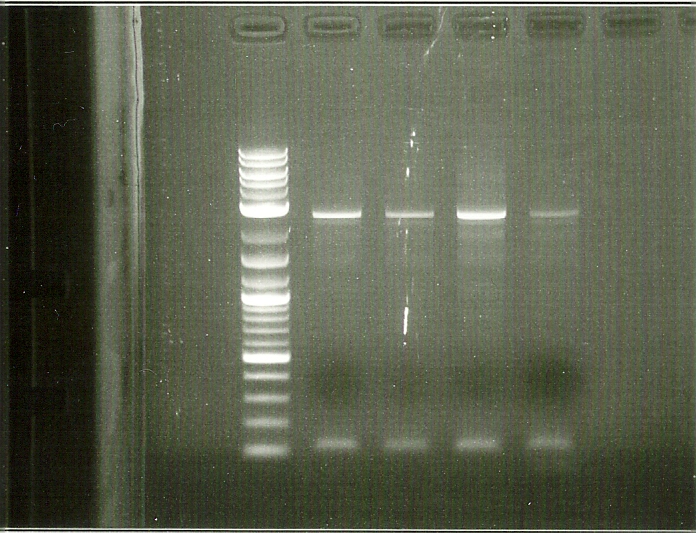
2nd round PCR Gel: Lanes from left to right: 2kb ladder, BTS1, crtE, crtI, crtYB
After transformation of our tagged genes, fluorescence microscopy was used to confirm the success of the transformation and expression of our tagged genes. Depicted in the fluorescence microscopy figure, the transformed yeast cells fluoresce red, which is evidence that our tagged mCherry (RFP) tagged genes are in fact present and being expressed in our yeast. Green fluorescence is seen in both wild type and transformed yeast, which is due to the autofluorescence inherent to beta carotene. In addition, the DIC (differential interference contrast) microscopy image shows the localization of our highly hydrophobic beta carotene molecules inside lipid droplets of the yeast cell.
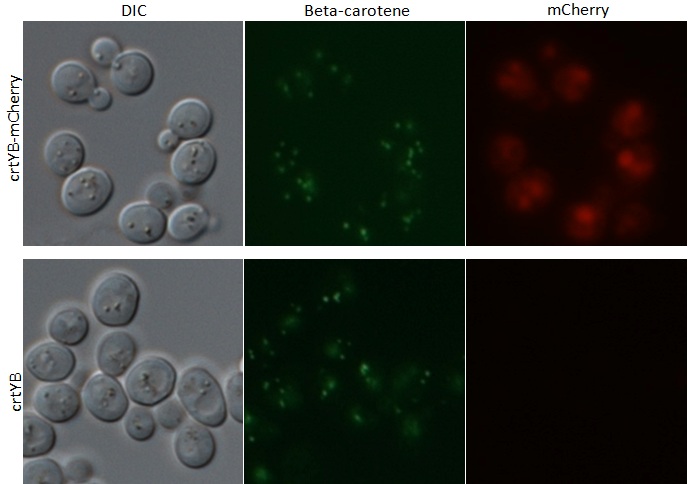
Microscopy Images from tagged crtYB-mCherry tagged yeast and crtYB control
References
1. High-Level Production of Beta-Carotene in Saccharomyces cerevisiae by Successive Transformation with Carotenogenic Genes from Xanthophyllomyces dendrorhous
René Verwaal,1, Jing Wang,1 Jean-Paul Meijnen,1 Hans Visser,1,Gerhard Sandmann,2 Johan A. van den Berg,1, and Albert J. J. van Ooyen1*
 "
"