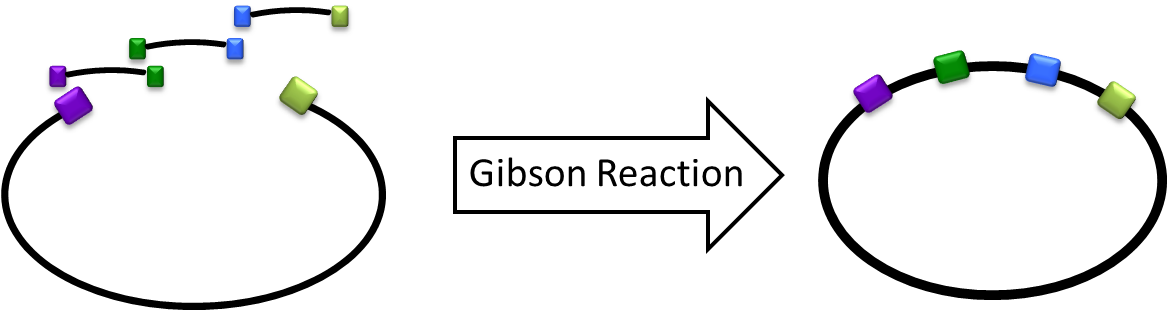Team:Washington/Magnetosomes/GibsonVectors
From 2011.igem.org
| Line 23: | Line 23: | ||
<br> | <br> | ||
| - | === What about this year? === | + | ===What about this year?=== |
| + | Seeing that this is a very efficient method to do cloning, we continued to make improvements to the methods and created a '''Gibson Assembly Toolkit'''! <br> <br> | ||
| - | + | [[File:Washington_iGEM2011_how_to_make_vector.png|left|thumb|450px]] | |
| - | + | ||
| - | + | ||
<br> | <br> | ||
| - | + | ===IGEM 2011--Creation of 5 plasmid vectors=== | |
| - | + | Our new vectors for Gibson assembly follow the naming convention of pGA. To make our pGA vectors, we first amplified the backbones and the pLac GFP insert respectively. Then we performed a Gibson reaction to combine them together to make the pGA vectors. | |
| - | To make our pGA vectors, we first amplified the backbones and the pLac GFP insert respectively. Then we performed a Gibson reaction to combine them together to make the pGA vectors. | + | |
| - | + | ||
| - | + | ||
All togeher, we created 5 pGA vectors and submitted them to the registry: | All togeher, we created 5 pGA vectors and submitted them to the registry: | ||
Revision as of 00:24, 24 September 2011
About Gibson Cloning/Assembly
Gibson Cloning/Assembly is a novel synthetic biology tool that allows multiple gene-inserts during a single isothermal reaction that is used for assembling overlapping DNA fragments. This method is gaining popularity as it tends be more efficient, saving a great amount of time during the cloning process. Overall, Gibson cloning allows teams to built more complex circuits with ease.
What happened last year?
The Gibson Cloning method is definitely not a new method to be introduced to the iGEM community.
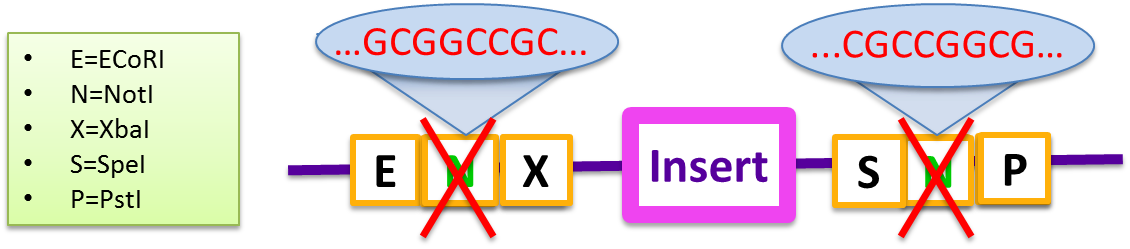
In 2010, the Cambridge iGEM team created the RFC 57 document which outlines a protocol for Gibson Assembly using Standard BioBricks that would allow multiple gene inserts during a single cloning event. However, while creating our Magnetosome Toolkit, we found that this BioBrick standard was incapable of producing high yields of Gibson products for even two-gene assemblies.
The primary problem with this standard pSB vector is the self-complementary of the two NotI sequences embedded in the BioBrick prefix and suffix. These sequences prevent gene inserts from being incorporated efficiently, and do not produce a high yield of the Gibson product even in two-fragments assembly.
- E=ECoRI, N=NotI, X=XbaI, S=SpeI, P=PstI
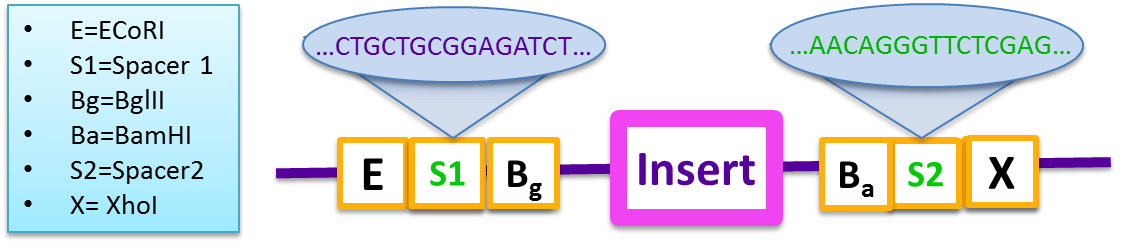
Therefore, to overcome this problem, our 2011 team developed a “gibson reaction compatible” prefix and suffix that were based on BglBrick standards. These vectors increase the cloning efficiency of large-scale gene assemblies and are also compatible with iGEM standard BioBrick parts.
- E=ECoRI,S1=Spacer 1, Bg=Bgl S2=Spacer2, P=PstI
What about this year?
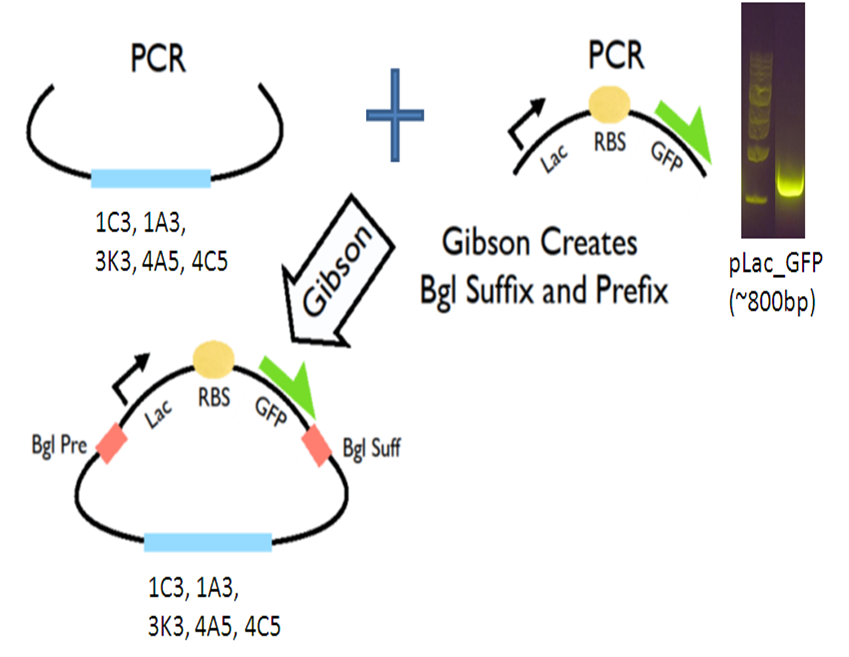
Seeing that this is a very efficient method to do cloning, we continued to make improvements to the methods and created a Gibson Assembly Toolkit!
IGEM 2011--Creation of 5 plasmid vectors
Our new vectors for Gibson assembly follow the naming convention of pGA. To make our pGA vectors, we first amplified the backbones and the pLac GFP insert respectively. Then we performed a Gibson reaction to combine them together to make the pGA vectors.
All togeher, we created 5 pGA vectors and submitted them to the registry:
- 2 High copy extraction/cloning vectors
- pGA1A3, pGA1C3
- 1 medium copy expression vector
- pGA3K3
- 2 low copy expression vectors
- pGA4A5, pGA4C5
As listed above, each of our vectors have varying copy numbers, antibiotic resistances, and purposes within the magnetosome gene assembly. However, they all appear to be very efficient will multiple gene inserts.
References
1.
 "
"