Team:Paris Bettencourt/GFP diff
From 2011.igem.org
| Line 6: | Line 6: | ||
<p>The plated bacteria were then observed through fluorescent microscopy. After a while (between 15 minutes and 2 hours), a transfer of GFP can be observed from the <i>gfp +</i> cells towards the <i>gfp-</i> cells. This cell-to-cell communication was previously unheard of and the original paper <a href="http://bms.ucsf.edu/sites/ucsf-bms.ixm.ca/files/marjordan_06022011.pdf">[1]</a> strongly suggest that the so-called nanotubes observed through electronic microscopy by the authors is the reason of this transfer.</p> | <p>The plated bacteria were then observed through fluorescent microscopy. After a while (between 15 minutes and 2 hours), a transfer of GFP can be observed from the <i>gfp +</i> cells towards the <i>gfp-</i> cells. This cell-to-cell communication was previously unheard of and the original paper <a href="http://bms.ucsf.edu/sites/ucsf-bms.ixm.ca/files/marjordan_06022011.pdf">[1]</a> strongly suggest that the so-called nanotubes observed through electronic microscopy by the authors is the reason of this transfer.</p> | ||
<h2>Our experiment</h2> | <h2>Our experiment</h2> | ||
| - | <p>It took | + | <p>It took several weeks to repeat the Dubey and Ben-Yehuda experiments to obtain a reliable microscopy observations. An important optimisation step in our protocol was done thanks to Ben-Yehuda's advice to focus on the creation of the monolayer. Plating the cells properly is actually more important than the actual concentration of the liquid mix.</p> |
<p>We began our experiments with a PY79 strain (as in the paper) but found quickly that our fluorescent version of this strain was rather weak. Hence starting from late august we chose to work with a 3610 strain which showed stronger fluorescence .</p> | <p>We began our experiments with a PY79 strain (as in the paper) but found quickly that our fluorescent version of this strain was rather weak. Hence starting from late august we chose to work with a 3610 strain which showed stronger fluorescence .</p> | ||
<p>Finally, after several tries, we managed to <em>reproduce the result expected: GFP diffusion between cells!</em></p> | <p>Finally, after several tries, we managed to <em>reproduce the result expected: GFP diffusion between cells!</em></p> | ||
Revision as of 13:34, 20 September 2011

GFP diffusion
The original experiment
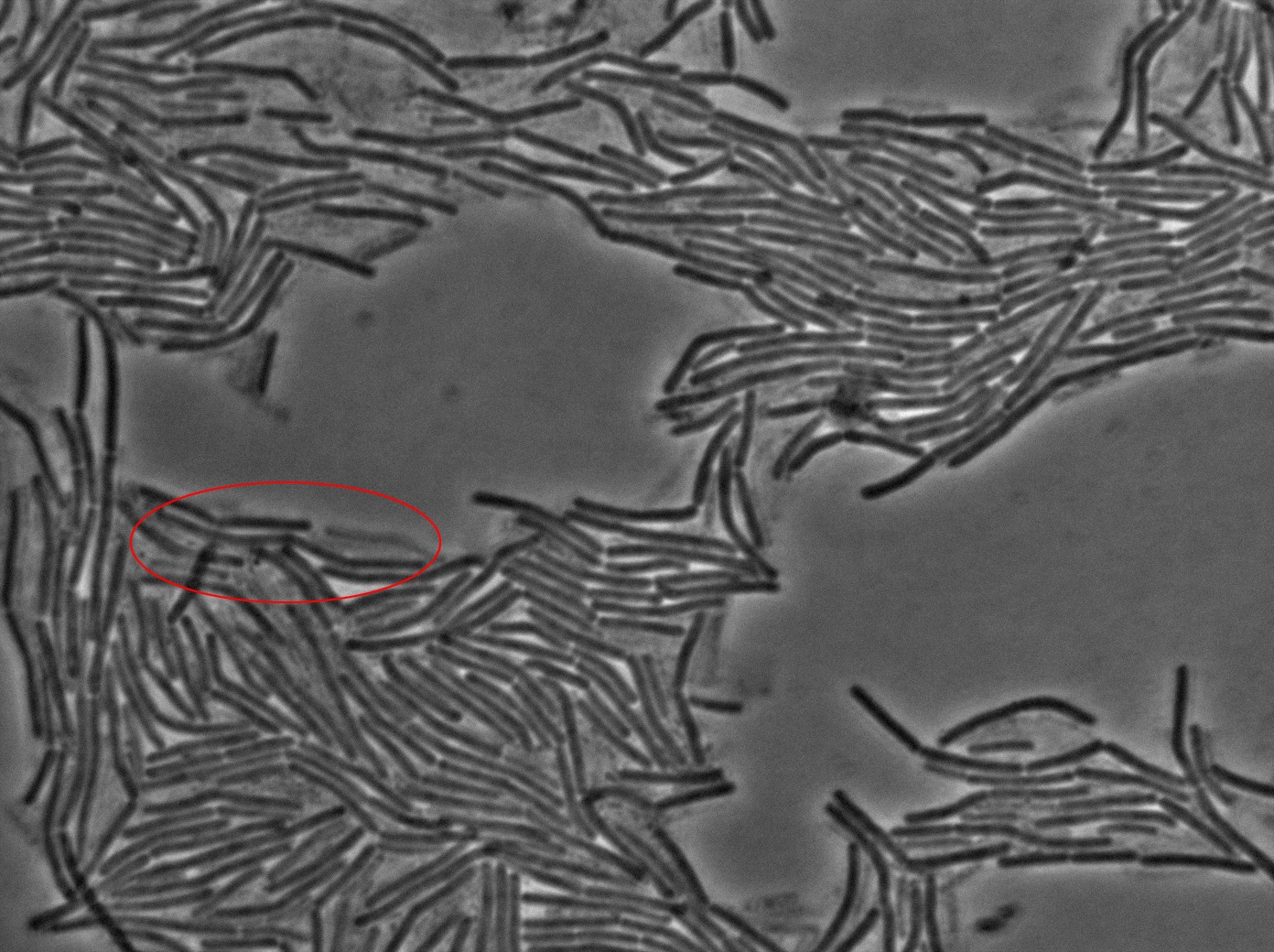
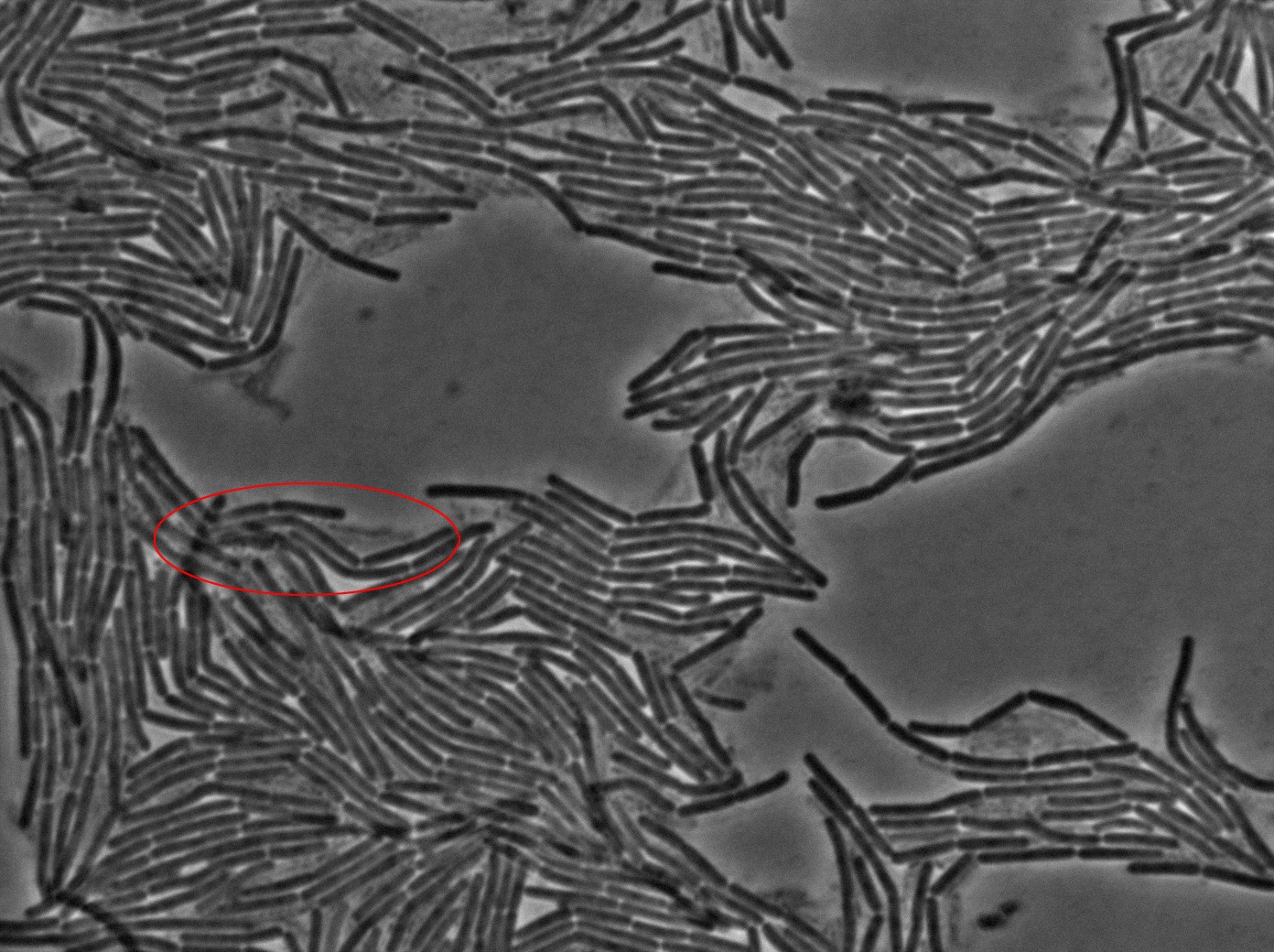
The keystone experiment of the Dubey and Ben-Yehuda paper [1] is simple. They used one strain of gfp + B.subtilis and one gfp - B.subtilis strain. The two strains of B.subtilis were put together (ratio 1:1) on an LB-agarose (1.5%) plate in exponantial phase. The goal is to have a monolayer of densely packed cells.
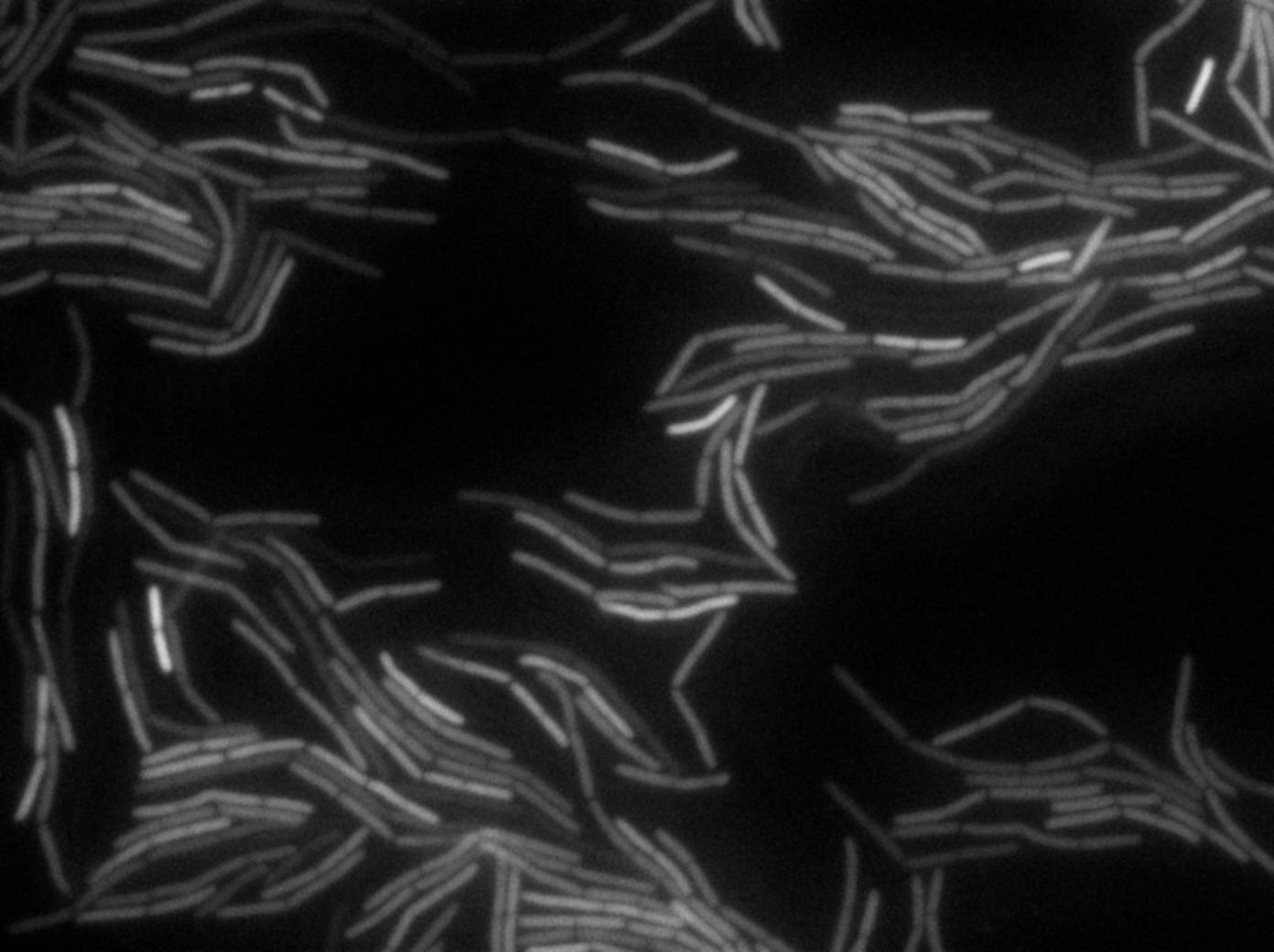
The plated bacteria were then observed through fluorescent microscopy. After a while (between 15 minutes and 2 hours), a transfer of GFP can be observed from the gfp + cells towards the gfp- cells. This cell-to-cell communication was previously unheard of and the original paper [1] strongly suggest that the so-called nanotubes observed through electronic microscopy by the authors is the reason of this transfer.
Our experiment
It took several weeks to repeat the Dubey and Ben-Yehuda experiments to obtain a reliable microscopy observations. An important optimisation step in our protocol was done thanks to Ben-Yehuda's advice to focus on the creation of the monolayer. Plating the cells properly is actually more important than the actual concentration of the liquid mix.
We began our experiments with a PY79 strain (as in the paper) but found quickly that our fluorescent version of this strain was rather weak. Hence starting from late august we chose to work with a 3610 strain which showed stronger fluorescence .
Finally, after several tries, we managed to reproduce the result expected: GFP diffusion between cells!

You can find out more about this exciting day on the Notebook.
Regarding the microscopy protocol you can refer to this page.
You can find the video we made on this link.
Conclusions of our experiment
- We have evidences of a cell-to-cell GFP transfer. This is probably a non-specific transfer (as GFP is not a natural protein for B.subtilis) and according to the Dubey and Ben-Yehuda paper [1], we can strongly syspect that it is done through nanotubes.
- Reproducing the original experiment is difficult. We were able to observe the expected behaviour in very few cells. This means either that the transfer is very rare or that we need to improve the preparation of the slide.
- We used a different strain (3610 instead of PY79). This means the transfer is not observed only in PY79.
- We proved that we will be able to see the results of our designs once we test them.
 "
"