Team:Caltech/Week 3
From 2011.igem.org
| Line 104: | Line 104: | ||
Redo PCR of [http://partsregistry.org/Part:BBa_R0010 R0010] and [http://partsregistry.org/Part:BBa_K123000 K123000]<br/> | Redo PCR of [http://partsregistry.org/Part:BBa_R0010 R0010] and [http://partsregistry.org/Part:BBa_K123000 K123000]<br/> | ||
Analyze sequence of ER, design new primer for continued sequencing<br/> | Analyze sequence of ER, design new primer for continued sequencing<br/> | ||
| + | Prepare overnight cultures of K145001 colonies for sequencing and overnight cultures of other biobrick plates for creating glycerol stocks<br/> | ||
Plan experiments using pNT001 and pNT002</p> | Plan experiments using pNT001 and pNT002</p> | ||
===Results=== | ===Results=== | ||
| Line 144: | Line 145: | ||
<p>Miniprep 5 K145001 cultures and send them off for sequencing before noon | <p>Miniprep 5 K145001 cultures and send them off for sequencing before noon | ||
Make glycerol stocks of mCherry ([http://partsregistry.org/Part:BBa_J06702 J06702]), Lac Promoter ([http://partsregistry.org/Part:BBa_R0010 R0010]), double terminator ([http://partsregistry.org/Part:BBa_B0014 B0014] and [http://partsregistry.org/Part:BBa_B0015 B0015]) and Tet Promoter ([http://partsregistry.org/Part:BBa_R0040 R0040])<br/> | Make glycerol stocks of mCherry ([http://partsregistry.org/Part:BBa_J06702 J06702]), Lac Promoter ([http://partsregistry.org/Part:BBa_R0010 R0010]), double terminator ([http://partsregistry.org/Part:BBa_B0014 B0014] and [http://partsregistry.org/Part:BBa_B0015 B0015]) and Tet Promoter ([http://partsregistry.org/Part:BBa_R0040 R0040])<br/> | ||
| - | + | Repeat PCR of B0014 and perform PCR to linearize backbone vector <br/> | |
| - | + | Digest PCR products with DpnI and purify<br/> | |
| - | + | Make gibson mix, run gibson reaction and transform<br/> | |
| - | + | ||
For gels tomorrow: Use Joe's sybr safe.</p> | For gels tomorrow: Use Joe's sybr safe.</p> | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
Revision as of 17:59, 30 June 2011
{{Team:Caltech/templateheader| Content=
June 26
Start overnight cultures of T7 polymerase (K145001), mCherry (J06702), Lac Promoter (R0010), double terminator (B0014 and B0015) and Tet Promoter (R0040)
June 27
Miniprep of T7 polymerase (K145001), mCherry (J06702), Lac Promoter (R0010), double terminator (B0014 and B0015) and Tet Promoter (R0040) and submit for sequencing
Transfer 0.5 mL aliquots of BPA and 5 mL aliquots 17a-ethynylestradiol cultures from June 23 to fresh minimal media
Checked DDT and nonylphenol cultures, will wait until tomorrow to transfer
Ethanol precipitation of DNA extractions from LA river samples following protocol
Streak out cells from fosmid kit
Prepare antibiotic stocks (Ampicillin, Chloramphenicol)
Results
Miniprep
| Part | Concentration(ng/ul) |
|---|---|
| B0014 | 230.5 |
| B0015 | 254.1 |
| J06702 | 301.3 |
| K145001 | 205.6 |
| R0010 | 117.3 |
| R0040 | 156.8 |
We found growth in one of our BPA enrichment cultures, and slight growth in our ethinyl estradiol cultures. We transferred these vials today.
June 28
Send off continued forward sequencing for HER
PCR for Gibson assembly of PNT001 and PNT002
Gel and PCR purification of PCR products
Analysis of sequencing results from yesterday
Transfer DDT and nonylphenol cultures to new media
Transform mCherry (J06702), Lac Promoter (R0010), double terminator (B0014 and B0015) and Tet Promoter (R0040)for creation of glycerol stocks
Results
Sequencing: All biobricks showed correct sequence except T7 Polymerase
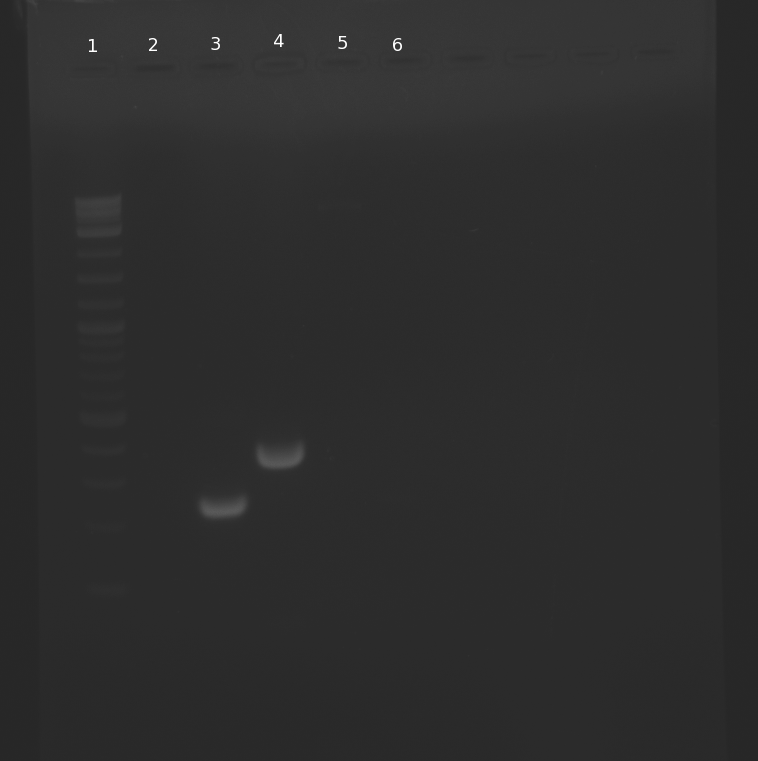
Ran a gel of the PCR products. Strangely, lanes 3 and 4 show no DNA. We will repeat the PCR of lanes 3-5 and Gibson assemble pNT001 (R0010, K123000, B0014) later in the week.
EtOH precipitation of Soil Extractions from June 21
| Tube/Location Number | Concentration(ng/ul) |
|---|---|
| 1 | 13.6 |
| 2 | 15.2 |
| 4 | 8.3 |
| 5 | 11.4 |
| 6 | 25.5 |
| 7 | 23.5 |
| 9 | 261.3 |
| 10 | 74.0 |
June 29
Redo PCR of R0010 and K123000
Analyze sequence of ER, design new primer for continued sequencing
Prepare overnight cultures of K145001 colonies for sequencing and overnight cultures of other biobrick plates for creating glycerol stocks
Plan experiments using pNT001 and pNT002
Results
Estrogen receptor (K123003) sequencing is back. Again, the translation is right, but the bases don't match. The stop codon is not in this read. We will keep sequencing.
B0014 was not amplified in PCR. We will PCR this part and primers again, as the PCR from June 28 has a rather low concentration to be used for Gibson Assembly.
Concentration of Purified PCR products from June 28 and June 29
| Tube Number | Concentration(ng/ul) |
|---|---|
| 6/28 3 | 15.2 |
| 6/28 4 | 49.1 |
| 6/28 5 | 42.0 |
| 6/28 6 | 112.0 |
| 6/29 1 | 112.7 |
| 6/29 2 | 168.0 |
June 30
Miniprep 5 K145001 cultures and send them off for sequencing before noon
Make glycerol stocks of mCherry (J06702), Lac Promoter (R0010), double terminator (B0014 and B0015) and Tet Promoter (R0040)
Repeat PCR of B0014 and perform PCR to linearize backbone vector
Digest PCR products with DpnI and purify
Make gibson mix, run gibson reaction and transform
For gels tomorrow: Use Joe's sybr safe.
 "
"