Team:Washington/Alkanes/Future/Modeling
From 2011.igem.org
Background
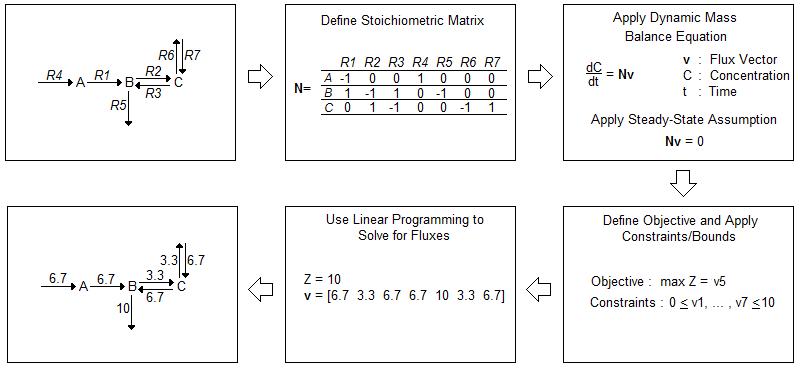
While in vivo analysis is invaluable, computational modeling can help direct experiments in a way that conserves time and resources. Current in silico models often rely on flux balance analysis (FBA) as an optimization algorithm. FBA provides a method for tracking fluxes within a biological system and “analyz[ing] biological networks in a quantitative manner” [1]. A flow chart of FBA methodology is depicted in Figure 1 [1].
FBA begins with the construction of an often complex biological network. After accounting for all fluxes within a system, FBA involves the construction of a stoichiometric matrix (N) where each column corresponds to a chemical reaction within the system and each row a molecular species. The matrix is then multiplied by a vector of internal and exchange fluxes (v), shown in Equation 1.
{1} dx/dt = Nv
Since FBA is a static model, the steady-state assumption, shown in Equation 2, applies. The overall equation simplifies to Equation 3.
{2} dx/dt = 0
{3} Nv = 0
Constraints are applied to the stoichiometric matrix to limit the number of possible solutions. These constraints often depend on the uptake rate and availability of nutrients within the media in which the cells are growing. A cell cannot take in a substrate if it is not available within its environment. After determining the constraints, an objective is defined, such as maximized protein production or maximized cell growth. Then, the fluxes that maximize the objective are mathematically calculated [1].
Methods
The modeling project began with an existing metabolic network for the MG1655 strain of E. coli (see Appendix). This model was obtained through the in silco organism database of professor and researcher Dr. Bernard Palsson, University of California: San Diego [2]. The alkane producing reactions were then added to the metabolic network. These reactions were simplified for the FBA system in order to minimize the number of new metabolites, and thus minimize the number of dead ends in the network. The simplified reactions were…
SIMPLIFIED REACTIONS
To then limit the number of solutions, constraints were applied to the system. The first of which concerned the uptake rate of glucose. For this value, we referred to a PNAS article, where researchers had already determined the needed rate for E. coli cells growing in a similar media [3]. Then, using the exact media composition used for this process, the exchange rate of thiamine, between the extracellular space and the cytoplasm, was adjusted such that the uptake rate of glucose by mass was equal to the uptake of thiamine by mass. Linear programming, the optimization process used by FBA, was then applied to determine the solution of fluxes for this system. With the model working, the next step was to apply modifications to optimize alkane production. Initial data showed that with the objective of the cell being growth, the in silico fluxes through the alkane producing pathway were zero. We propose that this observation could be explained in one of two ways. Either due to the hydrophobic nature of the alkane, it makes its way out of the cell into the surrounding media when produced. Thus, it does not contribute to the biomass of the cell, which is the overall objective of FBA. Also, an FBA does not capture the random probability of Acyl-ACP contacting Acyl-ACP reductase. This zero fluxes may also be due to the scale set by the biomass function. The growth of the cell, compared to the production of alkane, is very large. Since this system is centered about this growth, the FBA algorithm may consider the flux through the alkane producing pathway insignificant. This observation resulted in all following modifications focusing on maximizing production of Acyl-ACP to thus increase the probability of the metabolites entering the alkane producing pathway. Then using GDLS, a MATLAB function that suggests possible gene knockouts to maximize a certain flux, it was determined which knockouts would optimize production of Acyl-ACP. This process proposed the deletion of gene B1249, which encodes for cardiolipin synthase 1. The in silico deletion of this gene forced reactions that involved catabolizing of Acyl-ACP into smaller carrier proteins to zero. The deletion also significantly decreased the biomass function, which suggests further revisions should be made.
Future Modifications
References
Appendix
1. E. Coli metabolic network Media: Ec_iJR904_GlcMM.txt
2. MATLAB code for FBA Media: Matlab_Script.m
 "
"