Team:Kyoto/Digestion
From 2011.igem.org
Contents |
Project Digestion
Introduction
Streptomyces is a kind of prokaryotic bacteria which decompose bodies in nature[1]. We extract chitinase gene from this bacterium and introduce into Escherichia coli. Secretion-signal sequences are included in this gene so that the protein coded by them will go out without occurring cell lysis. After assembling all genes, we examined the activity of this enzyme in both quantitative ways.
Method
Construction
We created following construction to allow secretion of chitinase, chiA1. This gene is regulated by lactose promoter, BBa_R0011. We used Streptmyces’s RBS into this constructions, because reference article [1] used that to allow E.coli to secrete the protein.
Assay
We performed the quantitative assay, 3,5-Dinitrosalicylic acid assay (DNS method). This assay takes a little time and costs a little money.
DNS method is based on this fact: 3,5-dinitorosalicylic acid (DNS) is changed into 3-amino- 5-nitorosalicylic acid by reducing saccharide in reaction solution and the absorbance of this liquid increase in direct proportion to the amount of reducing sugar.
If chitinase is secreted in media by E.coli, chitin added in media will be decomposed to, for example, N-acetylglucosamin,that is ,reducing sugar. Therefore, The absorbance of the media added DNS will increase.
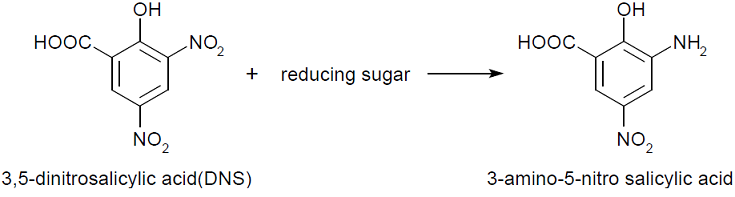
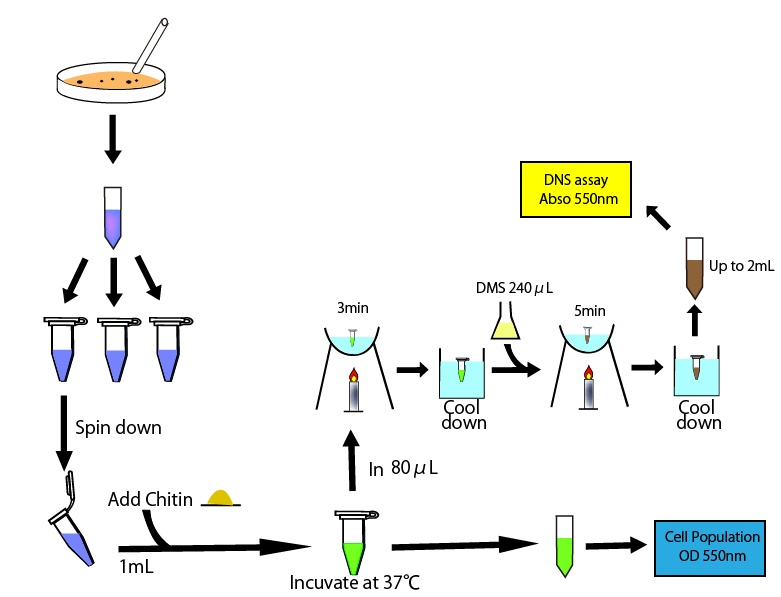
To carry out this assay, we thought four following dates would be required. These are
- the relationsip to reducing sugar concntration and absorbance 550
- the assesment of the component in media which can effect absobance 550
- the assesment of effect of E.coli in media
- the effect of chitinase
Procedure
The results of this measurement and the fig 1 graph enabled us to calculate the amount of digested chitin, showing the relative activity of chitinase.
preliminary experiments
1-1:build a standard curve
| Sample | glucose solution(various concentration from 0.20 mM to 1.60 mM) |
| Blank | mixed liquid (240μℓDNS reagent plus 1760㎕distilled water ) |
- added 240㎕ DNS reagent to 80㎕sample
- heated it for 5 min in boiling water and then cooled it in water.
- added it distilled water by 2ml
- measured absorbance in 550nm
1-2:evaluate the affection of medium
we examined the interruption based on the components of media(SOC, plas-grow and M9)
| Sample | each medium |
| Blank | mixed liquid (240μℓDNS reagent plus 1760㎕distilled water ) |
- added 240㎕ DNS reagent to 80㎕sample
- heated it for 5 min in boiling water and then cooled it in water.
- added it distilled water by 2ml
- measured absorbance in 550nm
1-3:measurement the effect of remainded E.coli
| Sample | each medium |
| Blank | mixed liquid (240μℓDNS reagent plus 1760㎕distilled water ) |
This assay was performed three times for each medium
- poured the medium cultured E.coli overnight 1.2 ㎕ into each five microcentritube.
- centrifuged them for 5 min at 5,000 rpm
- prepared new five microcentritube and move 800㎕ the supernatants into each of them.
- measure the OD550 of one tube(use fresh medium as a blank in following assays)
- one hour after, we measured OD 550 of other tube
- take 80㎕ supernatant and move it into new tube and then heated it for 3 min in boiling water and then cooled it in water.
- added 240㎕ DNS reagent and heated it for 3 min in boiling water and then cooled it in water.
- two, three, five hours after, we did above operation, taking supernatant, measured OD500, heating and cooling, applying DNS reagent and heating and cooling again.
- added all sample tube (containing 320㎕ solution) distilled water by 2ml and measure the absorbance of them in 550nm.
Result
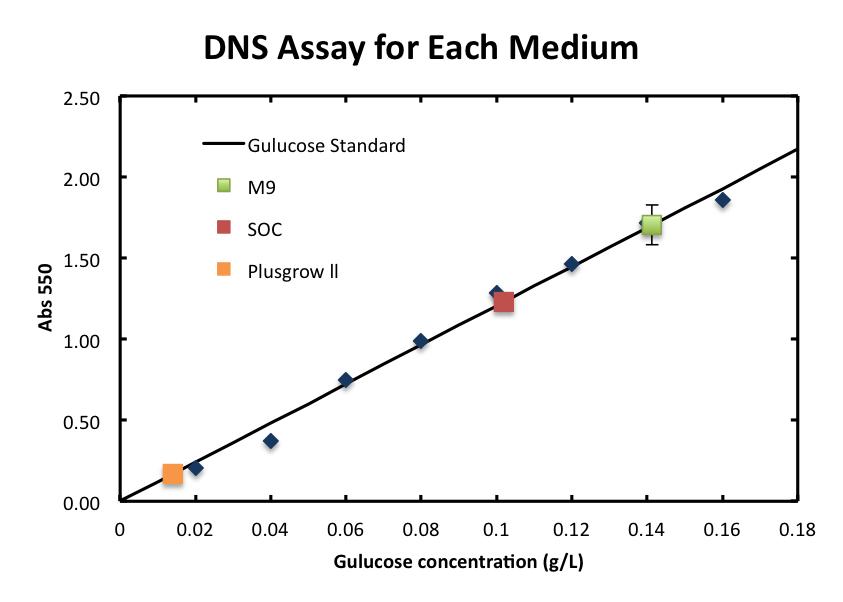
1. Standard Measurement for ChiA1.
- From the result, a strong correlation between glucose concentration and its A550 was observed.
2. Consideration of medium and growth of E.coli.
- We checked the influence of each medium to the DNS assay. Figure 2 shows the background absorbance of each medium. The absorbance of M9 was 1.7±0.1, SOC was1.227±0.007, and Plusgrow Ⅱ was 0.17±0.02.
Discussion
From result of preliminary experiments, we found several problems.
・the affection of medium itself
The components of each medium also reduced 3,5-dinitorosalicylic acid and would cause error in the assay.
・interruption of remaining E.coli.
Even though we use the centrifugal supernatant, there was still some E.coli. we found they could interrupt data because they would decompose reducing sugers.
To overcome these barriers, we decided detail plan of our assay.
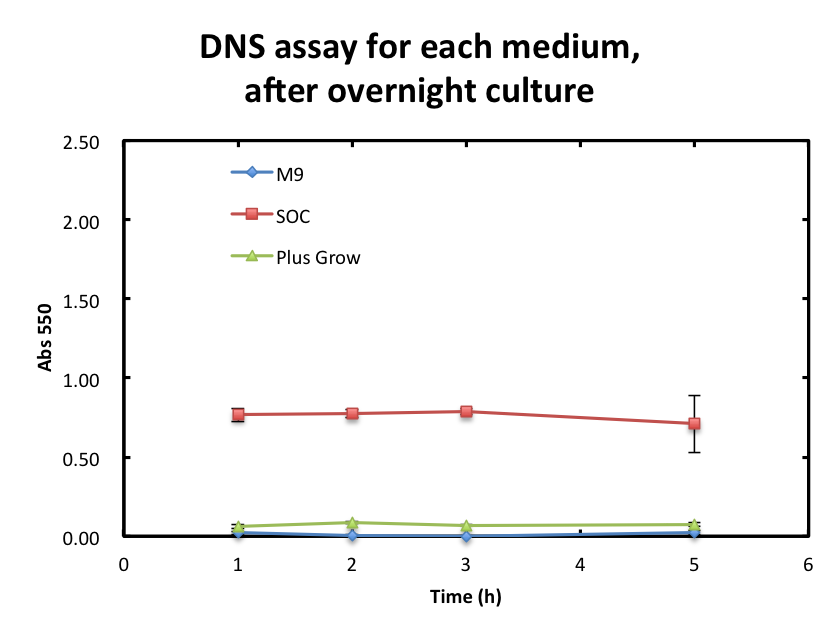
From the result fig 3, SOC medium cultured E.coli overnight would still include too much amount of reducing materials and, from fig 3. plas-grow enabled reminded E.coli to grow rapidly. However, as for M9 medium,
From result of preliminary experiments, we found several problems.
・the affection of medium itself
The components of each medium also reduced 3,5-dinitorosalicylic acid and would cause error in the assay.
・interruption of remaining E.coli.
Even though we use the centrifugal supernatant, there was still some E.coli. we found they could interrupt data because they would decompose reducing sugers.
To overcome these barriers, we decided detail plan of our assay. From the result fig 3, SOC medium cultured E.coli overnight would still include too much amount of reducing materials and, from fig 3. plas-grow enabled reminded E.coli to grow rapidly. However, as for M9 medium,
Reference
[1] “Actinobacteria.” Internet: http://en.wikipedia.org/wiki/Actinobacteria [Nov. 5, 2011]
[2] H. Ikeda, J. Ishikawa, A. Hanamoto, M. Shinose, H. Kikuchi, T. Shiba, Y. Sakaki, M. Hattori, S. Omura, “Complete genome sequence and comparative analysis of the industrial microorganism Streptomyces avermitilis.” Nat Biotechnol., vol. 21, no. 5 pp. 526-31, Apr. 2003
[3] S. Omura, H. Ikeda, J. Ishikawa, A. Hanamoto, C. Takahashi, M. Shinose, Y. Takahashi, H. Horikawa, H. Nakazawa, T. Osonoe, H. Kikuchi, T. Shiba, Y. Sakaki, M. Hattori, “Genome sequence of an industrial microorganism Streptomyces avermitilis: deducing the ability of producing secondary metabolites.” Proc Natl Acad Sci U S A. vol. 98, no. 21 pp. 12215-20, Oct. 9
[4] H. Sakuzou, ”還元糖の定量法(生物化学実験法)” Kyoto University: Japan Scientific Soceties Press
[5] S. Kongruang, M. J. Han, C. I. Breton, M. H. Penner, “Quantitative Analysis of Cellulose-Reducing Ends.” Appl Biochem Biotechnol. Vol. 113, no. 116 pp. 213-31, Spring 2004
 "
"