Team:Kyoto/Digestion
From 2011.igem.org
(→Assay) |
(→Project Digestion) |
||
| (152 intermediate revisions not shown) | |||
| Line 4: | Line 4: | ||
= '''Project Digestion''' = | = '''Project Digestion''' = | ||
| - | == '''Introduction''' == | + | == '''1. Introduction''' == |
| + | Insect bodies are covered with hard integument mainly composed of chitin. To decompose the integument, we used ChiA gene, which encodes secreted chitinase. In order to measure the chitinase activity of the culture supernatant, we evaluated the effects of the medium and cell growth.<br> | ||
| - | |||
| - | + | ChiA gene we used is derived from ''Streptomyces avermitilis''. ''S. avermitilis'' is a kind of prokaryotic bacteria and they produce chitinase.[2][3] We extracted this gene from ''S. avermitilis'' and introduced it into ''Escherichia coli''. This gene also encodes a secretion-signal sequence so that the chitinase will be secreted without the cell lysis of the ''E.coli''.<br> | |
| - | + | ||
| - | |||
| - | |||
| - | + | For characterizing the chitinase activity, we used the DNS assay, which is the method to determine the quantity of reducing sugar. In our assay, we determine the quantity of N-acetylglucosamine in a culture supernatant. Since the other materials in the supernatant can react with DNS, it was needed to examine if this assay is available for the quantitative characterization of chitinase activity. We also made a model to assess the enzyme activity in the supernatant from the pre-experiment.<br> | |
| - | + | =='''2. Method'''== | |
| - | + | ==='''Construction'''=== | |
| - | + | ||
| - | + | ||
| - | + | We designed the following construction to enable the secretion of the chitinase, ChiA1. This gene is regulated by a strong lactose promoter, BBa_R0011. We used ''Streptmyces''’s RBS into this construction, because in reference article [1] that RBS is used to allow ''E.coli'' to secrete the protein.<br/> | |
| - | + | [[File:Digestion_Constrctin1.png]]<br/> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | ===''' | + | ==='''Assay'''=== |
| - | + | We performed 3,5-Dinitrosalicylic acid assay (DNS assay), because this assay takes less time, costs a less money and is used in the previous article measuring chitinase activities [1]. DNS assay is based on this fact: Being boiled, 3,5-dinitorosalicylic acid (DNS), whose color is yellow, reacts with reducing saccharide by boiling and changes into 3-amino-5-nitorosalicylic acid, whose color is brawn. [4][5]<br> | |
| - | |||
| - | + | The more the amount of reducing sugar is, the more this coloring reaction proceeds. We can quantitatively evaluate the degree of this coloring reaction by measuring OD 550, because OD 550 is indirectly the amount of the reducing sugar. | |
| - | + | We will react DNS reagent with the media where chitin and E.coli introduced chitinase gene are added. If chitinase is secreted in media, chitin is decomposed into reducing sugar, for example, N-acetylglucosamin and coloring reaction proceeds. Therefore, by measuring OD 550, we can indirectly measure chitinase activities. <br> | |
| - | + | [[File:Kyoto-digestion-DNSassay1.jpg]]<br/> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| + | =='''3. Result'''== | ||
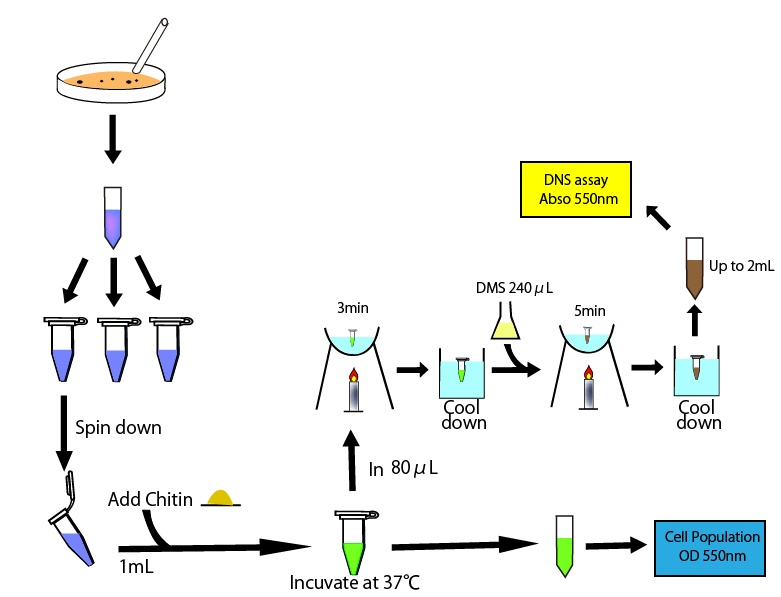
| - | + | This figure shows the overall of DNS assay | |
| - | + | [[File:キチナーゼ.PNG]] | |
| - | # | + | The detail of these experiment was written in <html><a href="/Team:Kyoto/Measurement#Measurement">Protocol</a></html> |
| - | + | ||
| - | |||
| - | + | To measure chitinase activity by DNS assay, we thought to need four following things to get accurate result. We need | |
| + | #to plot the relationship of reducing sugar concentration and OD 550 | ||
| + | #to evaluate the impact of components of media on OD 550 | ||
| + | #to evaluate the impact of remeineded E.coli in media on OD 550 | ||
| + | #to measure chitinase activity<br> | ||
| - | |||
| - | |||
| - | + | ==='''3-1. Standard Measurement for ChiA1.'''=== | |
| - | + | In order to know the relationship between OD550 and the amount of reducing sugar, we prepared the standard curve by using glucose as standard. It is known that the correlation between glucose concentration and the absorbance can be plotted in a linaer way.<br> | |
| - | + | From the result, a strong correlation between glucose concentration and its OD550 was observed. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
[[File:Kyoto-ChiA1Standard0925.png|thumb|center|350px|Fig.1: Absorbance550 vs. glucose concentration. ''r''<sup>''2''</sup>=0.98936.]] | [[File:Kyoto-ChiA1Standard0925.png|thumb|center|350px|Fig.1: Absorbance550 vs. glucose concentration. ''r''<sup>''2''</sup>=0.98936.]] | ||
| - | 2. Consideration of medium and growth of ''E.coli''. | + | ==='''3-2. Consideration of medium and growth of ''E.coli''.'''=== |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | To get the accurate values of absorbance, it is needed to consider the residual ''E.coli'' in the supernatant. While the supernatant is incubated for several hours for the enzyme reaction, the cells can grow, produce chitinase and consume reducing sugar for their metabolism, which should influence to the result of DNS assay.<br><br> | |
| - | + | ||
| - | + | :'''3-2-1 The time-course change of OD 550 of the supernatant.''' | |
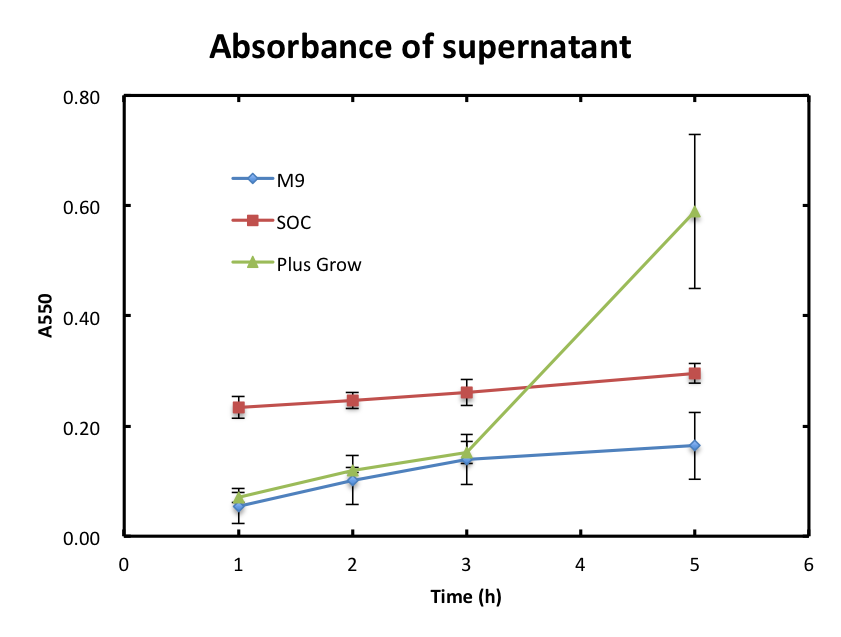
| + | :In this experiment, we checked if the cells were still observed in the supernatant and assayed the growth behavior of cells. Figure 2 shows the time-course change of OD 550 of supernatant. The absorbance values of M9 were hardly increased and they were smaller than 0.2 during the incubation. The absorbance values of SOC were also increased slowly, but were higher than those of M9. The absorbance of values of Plusgrow Ⅱ were increased obviously, especially between the 3h and 5h, and much higher than those of M9 and SOC.<br><br> | ||
| + | :From figure 2, the increase in absorbance of Plusgrow Ⅱ was thought to be due to the cell growth. So, we considered that the cells also existed in M9 and SOC medium though the significant cell growth couldn't observed. <br> | ||
| - | + | [[File:Kyoto-Absorbance_of_supernatant.png|thumb|center|400px|Fig.2: Absorbance of supernatant itself.]]<br> | |
| - | + | ||
| - | |||
| - | + | :'''3-2-2 The DNS assay of the supernatant.''' | |
| - | we | + | :In this experiment, we evaluated the influence of the cells and its growth to the DNS assay. In other words, we confirmed if cells consume reducing sugar and affect the result of DNS assay. Figure 3 shows the absorbance of each culture supernatant. The values of M9 and Plusgrow Ⅱ were almost zero during the incubation. The absorbance of SOC was observed, but there was little change during the incubation.<br><br> |
| - | + | :From figure 2 and 3, the cell growth didn't affect to the change of absorbance. About M9 and SOC medium, each medium contains only glucose as sugar source and it is considered that glucose was mostly consumed after overnight culture. This is the reason why the cell growth didn't observed. On the other hands, Plusgrow Ⅱ didn't contain reducing sugar, but the cell growth was observed. It can be considered that even if Plusgrow Ⅱ doesn't contain any reducing sugar, the cells consume polysaccharide for their metabolism.<br><br> | |
| - | + | [[File:Kyoto-DNS_assay_of_supernatant.png|thumb|center|400px|Fig.3: The influence of each culture supernatant to the DNS assay with time. Each supernatant was diluted 25 fold with water. Data points and error bars correspond to the mean and the standard deviation of three time experiments.]]<br> | |
| + | :'''3-2-3 The DNS assay of medium.''' | ||
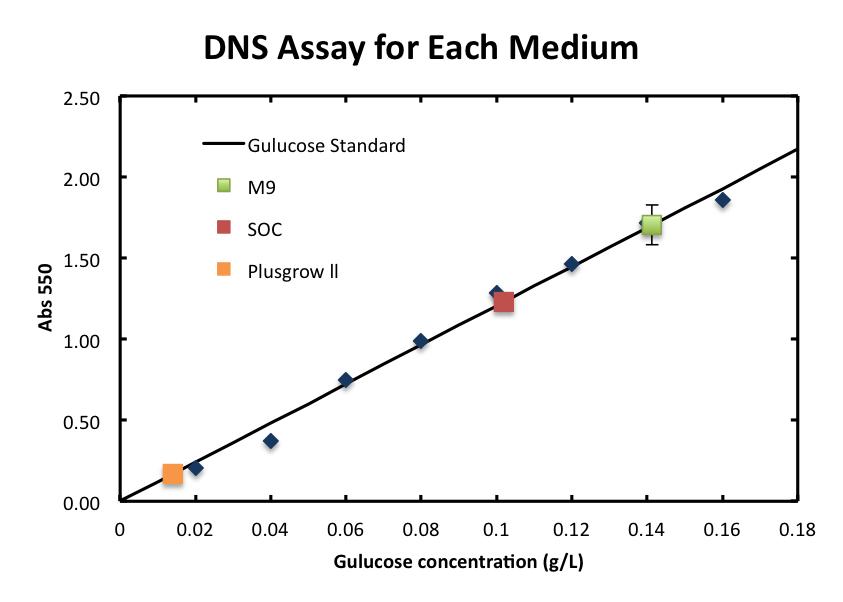
| + | :We checked the influence of each medium to the DNS assay. Figure 4 shows the background absorbance of each medium. The absorbance of M9 was 1.7±0.1, SOC was1.227±0.007, and Plusgrow Ⅱ was 0.17±0.02.<br><br> | ||
| - | + | :The data of Plusgrow Ⅱ supports the fact that little reducing sugar is contained in this medium. | |
| - | + | [[File:Kyoto-DNSassayforeachmedium.png|thumb|center|400px|Fig.4: The influence of each medium to the DNS assay. Each medium was diluted 25 fold with water. Data points and error bars correspond to the mean and the standard deviation of three time experiments.]] | |
| - | + | =='''4. Discussion'''== | |
| - | + | ==='''Plusgrow Ⅱ should be suit for the DNS assay in the first three hours.'''=== | |
| + | From three experiments: 3-2-1, 2, and 3, Plusgrow Ⅱ is good for characterizing the chitinase activity using the DNS assay in the first three hours. Firstly, cell number didn't increase so much in the first three hours. So, we can perform the chitinase assay that based on the premise that the enzyme concentration is nearly constant. Secondly, since ''E.coli'' can grow in the supernatant without having the influence to the result of DNS assay, the residual ''E.coli'' doesn't consume reducing sugar, and N-acetylglucosamine will be assayed quantitatively by DNS.<br><br> | ||
| - | + | =='''5.Achievment'''== | |
| + | We aimed at creating prefect chitinase constraction. However, We don't get this because of time shortage.<br> | ||
| + | Finally, we could creat following constraction.<br> | ||
| + | [[File:Digestion_Constraction2.png]] | ||
| - | + | Moreover, We succesed modeling of chitinase activity. | |
| - | + | <html><a href="/Team:Kyoto/Digestion/Modeling">Modeling</a></html><br> | |
=='''Reference'''== | =='''Reference'''== | ||
| Line 161: | Line 99: | ||
[3] S. Omura, H. Ikeda, J. Ishikawa, A. Hanamoto, C. Takahashi, M. Shinose, Y. Takahashi, H. Horikawa, H. Nakazawa, T. Osonoe, H. Kikuchi, T. Shiba, Y. Sakaki, M. Hattori, “Genome sequence of an industrial microorganism Streptomyces avermitilis: deducing the ability of producing secondary metabolites.” Proc Natl Acad Sci U S A. vol. 98, no. 21 pp. 12215-20, Oct. 9 | [3] S. Omura, H. Ikeda, J. Ishikawa, A. Hanamoto, C. Takahashi, M. Shinose, Y. Takahashi, H. Horikawa, H. Nakazawa, T. Osonoe, H. Kikuchi, T. Shiba, Y. Sakaki, M. Hattori, “Genome sequence of an industrial microorganism Streptomyces avermitilis: deducing the ability of producing secondary metabolites.” Proc Natl Acad Sci U S A. vol. 98, no. 21 pp. 12215-20, Oct. 9 | ||
| - | [4] H. Sakuzou, ”還元糖の定量法(生物化学実験法)” Kyoto University: Japan Scientific Soceties Press | + | [4] H. Sakuzou, ”還元糖の定量法(生物化学実験法)(''kangento no teiryoho'')” Kyoto University: Japan Scientific Soceties Press |
[5] S. Kongruang, M. J. Han, C. I. Breton, M. H. Penner, “Quantitative Analysis of Cellulose-Reducing Ends.” Appl Biochem Biotechnol. Vol. 113, no. 116 pp. 213-31, Spring 2004 | [5] S. Kongruang, M. J. Han, C. I. Breton, M. H. Penner, “Quantitative Analysis of Cellulose-Reducing Ends.” Appl Biochem Biotechnol. Vol. 113, no. 116 pp. 213-31, Spring 2004 | ||
Latest revision as of 03:59, 6 October 2011
Contents |
Project Digestion
1. Introduction
Insect bodies are covered with hard integument mainly composed of chitin. To decompose the integument, we used ChiA gene, which encodes secreted chitinase. In order to measure the chitinase activity of the culture supernatant, we evaluated the effects of the medium and cell growth.
ChiA gene we used is derived from Streptomyces avermitilis. S. avermitilis is a kind of prokaryotic bacteria and they produce chitinase.[2][3] We extracted this gene from S. avermitilis and introduced it into Escherichia coli. This gene also encodes a secretion-signal sequence so that the chitinase will be secreted without the cell lysis of the E.coli.
For characterizing the chitinase activity, we used the DNS assay, which is the method to determine the quantity of reducing sugar. In our assay, we determine the quantity of N-acetylglucosamine in a culture supernatant. Since the other materials in the supernatant can react with DNS, it was needed to examine if this assay is available for the quantitative characterization of chitinase activity. We also made a model to assess the enzyme activity in the supernatant from the pre-experiment.
2. Method
Construction
We designed the following construction to enable the secretion of the chitinase, ChiA1. This gene is regulated by a strong lactose promoter, BBa_R0011. We used Streptmyces’s RBS into this construction, because in reference article [1] that RBS is used to allow E.coli to secrete the protein.

Assay
We performed 3,5-Dinitrosalicylic acid assay (DNS assay), because this assay takes less time, costs a less money and is used in the previous article measuring chitinase activities [1]. DNS assay is based on this fact: Being boiled, 3,5-dinitorosalicylic acid (DNS), whose color is yellow, reacts with reducing saccharide by boiling and changes into 3-amino-5-nitorosalicylic acid, whose color is brawn. [4][5]
The more the amount of reducing sugar is, the more this coloring reaction proceeds. We can quantitatively evaluate the degree of this coloring reaction by measuring OD 550, because OD 550 is indirectly the amount of the reducing sugar.
We will react DNS reagent with the media where chitin and E.coli introduced chitinase gene are added. If chitinase is secreted in media, chitin is decomposed into reducing sugar, for example, N-acetylglucosamin and coloring reaction proceeds. Therefore, by measuring OD 550, we can indirectly measure chitinase activities.
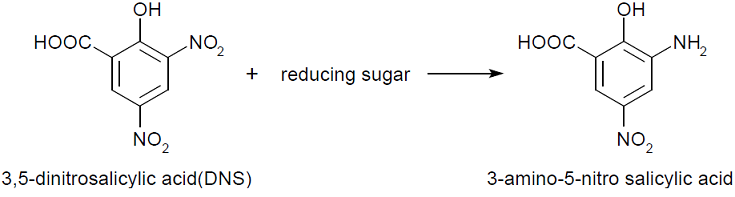
3. Result
This figure shows the overall of DNS assay
To measure chitinase activity by DNS assay, we thought to need four following things to get accurate result. We need
- to plot the relationship of reducing sugar concentration and OD 550
- to evaluate the impact of components of media on OD 550
- to evaluate the impact of remeineded E.coli in media on OD 550
- to measure chitinase activity
3-1. Standard Measurement for ChiA1.
In order to know the relationship between OD550 and the amount of reducing sugar, we prepared the standard curve by using glucose as standard. It is known that the correlation between glucose concentration and the absorbance can be plotted in a linaer way.
From the result, a strong correlation between glucose concentration and its OD550 was observed.
3-2. Consideration of medium and growth of E.coli.
To get the accurate values of absorbance, it is needed to consider the residual E.coli in the supernatant. While the supernatant is incubated for several hours for the enzyme reaction, the cells can grow, produce chitinase and consume reducing sugar for their metabolism, which should influence to the result of DNS assay.
- 3-2-1 The time-course change of OD 550 of the supernatant.
- In this experiment, we checked if the cells were still observed in the supernatant and assayed the growth behavior of cells. Figure 2 shows the time-course change of OD 550 of supernatant. The absorbance values of M9 were hardly increased and they were smaller than 0.2 during the incubation. The absorbance values of SOC were also increased slowly, but were higher than those of M9. The absorbance of values of Plusgrow Ⅱ were increased obviously, especially between the 3h and 5h, and much higher than those of M9 and SOC.
- From figure 2, the increase in absorbance of Plusgrow Ⅱ was thought to be due to the cell growth. So, we considered that the cells also existed in M9 and SOC medium though the significant cell growth couldn't observed.
- 3-2-2 The DNS assay of the supernatant.
- In this experiment, we evaluated the influence of the cells and its growth to the DNS assay. In other words, we confirmed if cells consume reducing sugar and affect the result of DNS assay. Figure 3 shows the absorbance of each culture supernatant. The values of M9 and Plusgrow Ⅱ were almost zero during the incubation. The absorbance of SOC was observed, but there was little change during the incubation.
- From figure 2 and 3, the cell growth didn't affect to the change of absorbance. About M9 and SOC medium, each medium contains only glucose as sugar source and it is considered that glucose was mostly consumed after overnight culture. This is the reason why the cell growth didn't observed. On the other hands, Plusgrow Ⅱ didn't contain reducing sugar, but the cell growth was observed. It can be considered that even if Plusgrow Ⅱ doesn't contain any reducing sugar, the cells consume polysaccharide for their metabolism.
- 3-2-3 The DNS assay of medium.
- We checked the influence of each medium to the DNS assay. Figure 4 shows the background absorbance of each medium. The absorbance of M9 was 1.7±0.1, SOC was1.227±0.007, and Plusgrow Ⅱ was 0.17±0.02.
- The data of Plusgrow Ⅱ supports the fact that little reducing sugar is contained in this medium.
4. Discussion
Plusgrow Ⅱ should be suit for the DNS assay in the first three hours.
From three experiments: 3-2-1, 2, and 3, Plusgrow Ⅱ is good for characterizing the chitinase activity using the DNS assay in the first three hours. Firstly, cell number didn't increase so much in the first three hours. So, we can perform the chitinase assay that based on the premise that the enzyme concentration is nearly constant. Secondly, since E.coli can grow in the supernatant without having the influence to the result of DNS assay, the residual E.coli doesn't consume reducing sugar, and N-acetylglucosamine will be assayed quantitatively by DNS.
5.Achievment
We aimed at creating prefect chitinase constraction. However, We don't get this because of time shortage.
Finally, we could creat following constraction.

Moreover, We succesed modeling of chitinase activity.
Modeling
Reference
[1] “Actinobacteria.” Internet: http://en.wikipedia.org/wiki/Actinobacteria [Nov. 5, 2011]
[2] H. Ikeda, J. Ishikawa, A. Hanamoto, M. Shinose, H. Kikuchi, T. Shiba, Y. Sakaki, M. Hattori, S. Omura, “Complete genome sequence and comparative analysis of the industrial microorganism Streptomyces avermitilis.” Nat Biotechnol., vol. 21, no. 5 pp. 526-31, Apr. 2003
[3] S. Omura, H. Ikeda, J. Ishikawa, A. Hanamoto, C. Takahashi, M. Shinose, Y. Takahashi, H. Horikawa, H. Nakazawa, T. Osonoe, H. Kikuchi, T. Shiba, Y. Sakaki, M. Hattori, “Genome sequence of an industrial microorganism Streptomyces avermitilis: deducing the ability of producing secondary metabolites.” Proc Natl Acad Sci U S A. vol. 98, no. 21 pp. 12215-20, Oct. 9
[4] H. Sakuzou, ”還元糖の定量法(生物化学実験法)(kangento no teiryoho)” Kyoto University: Japan Scientific Soceties Press
[5] S. Kongruang, M. J. Han, C. I. Breton, M. H. Penner, “Quantitative Analysis of Cellulose-Reducing Ends.” Appl Biochem Biotechnol. Vol. 113, no. 116 pp. 213-31, Spring 2004
 "
"