Team:Calgary/Project/Promoter/Final Data
From 2011.igem.org
Emily Hicks (Talk | contribs) |
|||
| Line 5: | Line 5: | ||
BODY = | BODY = | ||
<html> | <html> | ||
| - | < | + | <!-- --> |
| - | + | <h1>Introduction</h1> | |
| - | + | ||
| + | <p>In order to be able to utilize the electrochemical detection system, various response levels must be correlated to cell concentration. Because our output signal is the product of a reaction sequence, it would not be sufficient to only characterize the performance of one particular part operating alone. This novel approach to characterizing LacZ through the oxidation of chlorophenol red was done in conditions allowing all the necessary reactions to occur simultaneously - just as if used in a field-ready biosensor.</p> | ||
| + | |||
| + | <p>The final results of our data processing is shown in Figure 1 below. The calculations done to arrive at this final curve follow in Appendix A.</p> | ||
| + | |||
| + | </html>[[image:Calgary2011_100uLgraph.png|thumb|400px|center|<b>Figure 1</b> Rate of current change versus input voltage for a cyclic voltammetry assay. The electrochemical cell contained 100uL 0.5M CPRG, 1mL NaH2PO4 buffer, 100uL induced cells containing a LacZ reporter with an IPTG inducible promoter, and nanopure water up to 20mL.]]<html> | ||
| + | |||
| + | |||
| + | <h1>Appendix A - Calculations</h1> | ||
| + | <p>In order to construct our standard curve for current response vs. number of cells, several experiments were conducted with varying numbers of cells, with a constant amount of CPRG (100uL at 0.5M), NaH2PO4 buffer (1mL at 0.5M), and nanopure water (up to 20mL). The cell was left for 5 minutes before the experiment was run.</p> | ||
| + | <p>Figure 2 below shows experimental data for a cyclic voltammetry experiment done with 7.5*10^5 cells.</p> | ||
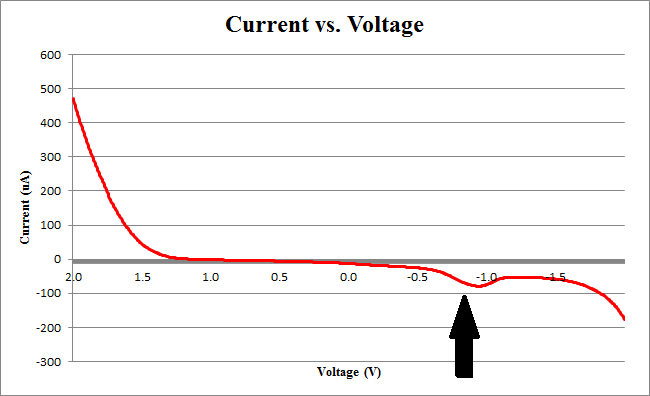
| + | </html>[[image:Calgary2011 100uL2.png|thumb|400px|center|<b>Figure 1</b> Current plotted against voltage during a cyclic voltammetry run.]]<html> | ||
| + | |||
| + | <p>We observe the characteristic dip indicative of CPR oxidation at approximately -0.71V, showing the presence of CPR in solution. Next, the time derivative of this curve will be taken, shown below.</p> | ||
| + | |||
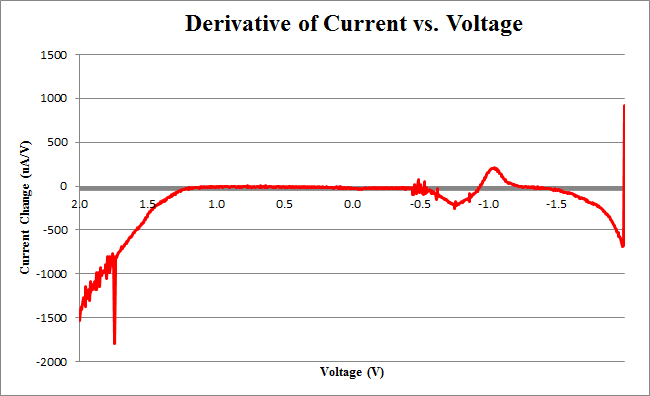
| + | </html>[[image:Calgary2011_100uLgraph.png|thumb|400px|center|<b>Figure 1</b> Rate of current change versus input voltage for a cyclic voltammetry assay. The electrochemical cell contained 100uL 0.5M CPRG, 1mL NaH2PO4 buffer, 100uL induced cells containing a LacZ reporter with an IPTG inducible promoter, and nanopure water up to 20mL.]]<html> | ||
| + | |||
| + | <p>Using Figure 3, we determine the maximum rate of change of current to occur at -0.755V, with a maximum of 258.45uA/V.</p> | ||
| + | |||
| + | <p>By repeating this procedure for the various tested cell levels, we </p> | ||
</html> | </html> | ||
}} | }} | ||
Revision as of 22:22, 28 September 2011









Characterization

Introduction
In order to be able to utilize the electrochemical detection system, various response levels must be correlated to cell concentration. Because our output signal is the product of a reaction sequence, it would not be sufficient to only characterize the performance of one particular part operating alone. This novel approach to characterizing LacZ through the oxidation of chlorophenol red was done in conditions allowing all the necessary reactions to occur simultaneously - just as if used in a field-ready biosensor.
The final results of our data processing is shown in Figure 1 below. The calculations done to arrive at this final curve follow in Appendix A.
Appendix A - Calculations
In order to construct our standard curve for current response vs. number of cells, several experiments were conducted with varying numbers of cells, with a constant amount of CPRG (100uL at 0.5M), NaH2PO4 buffer (1mL at 0.5M), and nanopure water (up to 20mL). The cell was left for 5 minutes before the experiment was run.
Figure 2 below shows experimental data for a cyclic voltammetry experiment done with 7.5*10^5 cells.
We observe the characteristic dip indicative of CPR oxidation at approximately -0.71V, showing the presence of CPR in solution. Next, the time derivative of this curve will be taken, shown below.
Using Figure 3, we determine the maximum rate of change of current to occur at -0.755V, with a maximum of 258.45uA/V.
By repeating this procedure for the various tested cell levels, we

 "
"