Team:Calgary/Project/Reporter
From 2011.igem.org









Reporter

What is a Reporter?
It isn't enough to just be able to detect something - after the presence of naphthenic acids are confirmed by our promoter, we require some way for our bacteria to report back results that can be interpreted by an observer. Over the course of the project, Team Calgary considered three different reporter systems - colorimetric, fluorescent, and electrochemical.
Why Electrochemical?
Oilsands tailings ponds samples are difficult to work with for several different reasons. First of all, tailings ponds samples are murky, and can have greatly varying compositions from pond to pond. It would be difficult to obtain accurate colorimetric data from these samples, and it would be similarly difficult to readily observe fluorescence. Because an electrochemical response does not rely on being able to see anything, Team Calgary determined the electrochemical reporter to be ideal for this purpose.
Electrochemical reporters offer several advantages over visually-determined reporters. The signal is robust - environmental factors such as the presence of pollutants or sample turbidity do not affect the signal. Every molecule has a different oxidation potential - because we know the exact oxidation potential for our analyte, we can easily tell the difference between our desired signal and background noise. Another advantage of an electrochemical response is that the data is immediately available in digital form. This allows for fast graphical interpretation of the data as well as maximum information collection from the data.
Electrical Reporter
This area should talk about the electrochemistry and explain the graphs
During the development of our electrochemical reporter system we spent a lot of time thinking about what hardware could be used and found out that a potentiostat is the best option. A potentiostat is the electronic hardware needed to control the three electrode system used to measure the data in our experiments. It keeps the working electrode at a constant potential with respect to the reference electrode by adjusting and measuring the current between the working electrode and counter electrode. We obtained our data using a professional lab grade potentiostat, but we also spent some time looking into how they are designed and managed to built our own prototype. While it is built on a breadboard and is bigger than the potentiostat we used for the measurements, it is a valid proof of concept that potentiostats can be built using electrical circuit components no more complicated than an operational amplifier.Testing the IPTG inducible LacZ Reporter
Background and Rationale
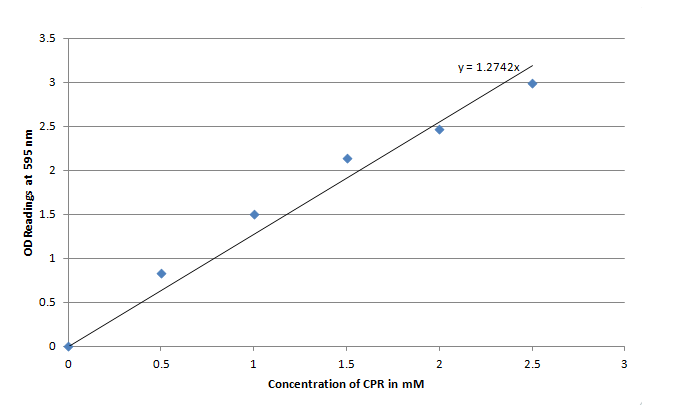
For the purposes of testing out the LacZ reporter as an electrochecmical detector it was important to first assess how well the reporter performed at cleaving the chlorophenol red β-D-galactopyranoside (CPRG) into Chlorophenol red (CPR). The LacZ gene that was chosen for this experiment as well as for the electrochemical testing was fused to an IPTG inducible promoter(LacI), or part I732901. Overall I732901 the purpose of the experiment was to ensure that the E. coli could be induced to produce β-galactosidase, which could then cleave chlorophenol red β-D-galactopyranoside (CPRG) to produce Chlorophenol red (CPR). Since CPR has a very dark characteristic red color at pH 7 the reaction wasto be kept at that constant pH using a phosphate buffer. This experiment was also expected to provide useful data for electrochemical side of things (i.e. oxidizing the CPR and measuring either the current or voltage) by determining what sort of time frame would be needed for the E. coli to produce a significant amount of CPR by cleavage of CPRG providing a guideline for future electrochemical testing.
Methods
E. coli carrying the plasmid were subcultured cultured until log phase then exposed to PBS buffer containing 3mM CPRG and 1mM IPTG for fixed lengths of time before measuring the absorbance of the solution. A detailed procedure are recorded in protocols section.
Results
Discussion of Results
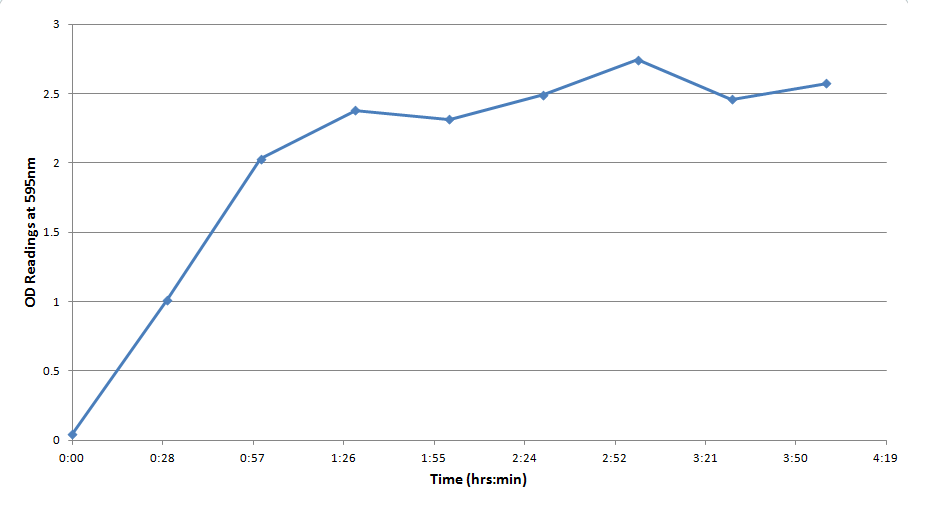
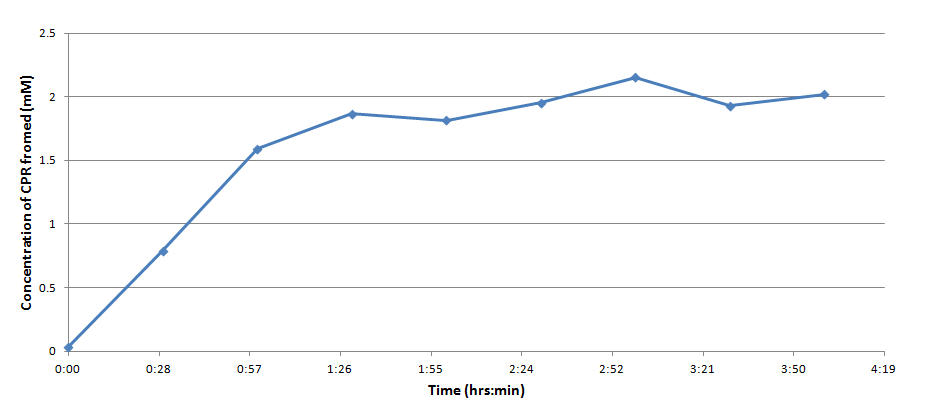
Figure 3 indicates that the concentration of CPR clearly increases over time in this mixture. Illustrating that the IPTG is successfully inducing the LacZ gene to produce β-galactosidase. This data will be useful to the electrochemical detector side of the project because they will now have data from which they can choose a time when they should begin oxidizing the CPR formed by the E. coli (by cleavage of CPRG).
One drawback experienced in this experiment was that the Victor plate reader which was used to take OD readings could not give data past an OD of ~3 for CPR absorption, meaning that even samples with an extremely high concentration of CPR would at best have an absorbance reading similar to a ~3 mM sample. Thus, this method was not particularly accurate for highly concentrated solutions of CPR. However the results did reveal that under the specified conditions about 1mM of CPR could be produced in around 30 minutes revealing that the system responded quite rapidly when induced with IPTG.

 "
"