Team:Paris Bettencourt/Experiments/T7 diffusion
From 2011.igem.org

T7 diffusion experiments
Design overview
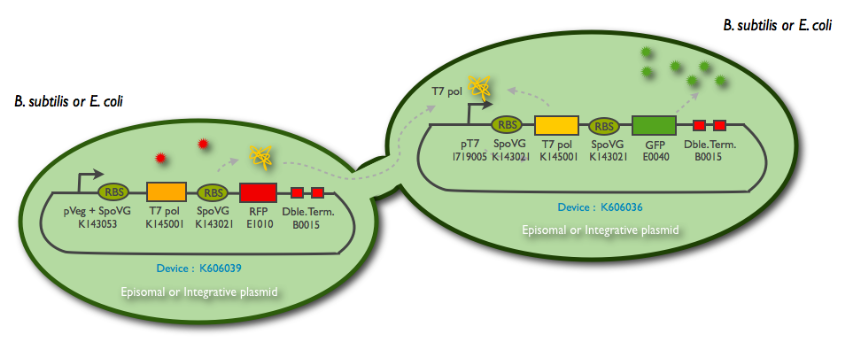
 Fig1: Schematic summary of the T7 diffusion device
Fig1: Schematic summary of the T7 diffusion device
More information on the design here: link
Parts
We synthetized the pHyperSpank promoter (<partinfo>BBa_K143055</partinfo>), but we were not able to incorporate it into our biggest construct. Instead, we cloned a Pveg promoter SpoVG (<partinfo>BBa_K143053</partinfo>) that has a constitutive expression.
The following schematic represents what we have indeed managed to clone:
This summary shows the parts we have characterized in E.coli and the ones we have sent to the registry.
Characterization of the T7 promoter
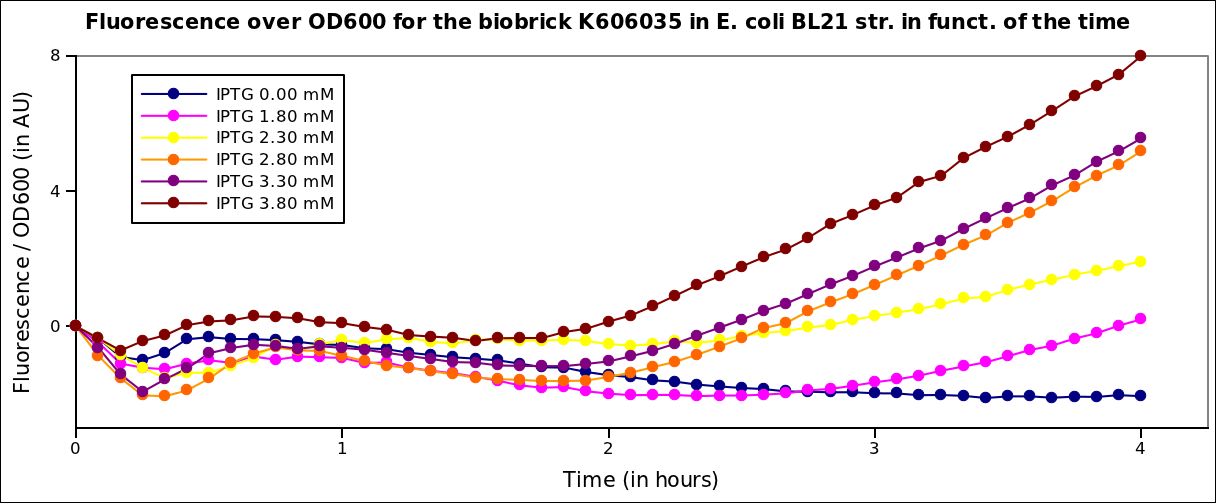
In order to characterize the pT7 promoter, we used the construct pT7-RBS-GFP-T7ter. This construct was transformed into BL21 strains expressing the T7 polymerase under IPTG induction.Fluorescence kinetics
The measurements have been carried out on a spectrophotometer at 37°C under transient shaking. The experiment lasted 4h, we tested several colonies and several IPTG concentrations. The OD at 600nm and the fluorescence of the GFP (exc: 470nm / meas:515 nm) was measured every 5 min and the ratio of the two was calculated.
All values were normalized by substracting the fluorescence/OD value of the well with 0 mM IPTG at time 0. The values given are in arbitrary units.
After 2 hrs of induction, we see a clear increase of the fluorescence proportional to the IPTG concentration (that is to say with the quantity of T7 polymerase induced in the cell). After 4 hrs, the expression of GFP under the pT7 is still not saturated
Here we plot the ratio of induction of the T7 polymerase dependant construct for the different concentrations of IPTG at a given time (4 hrs) taking the well with 0 IPTG at time 0 as the reference.
Characterization of the T7 signal amplification leakage in E. coli
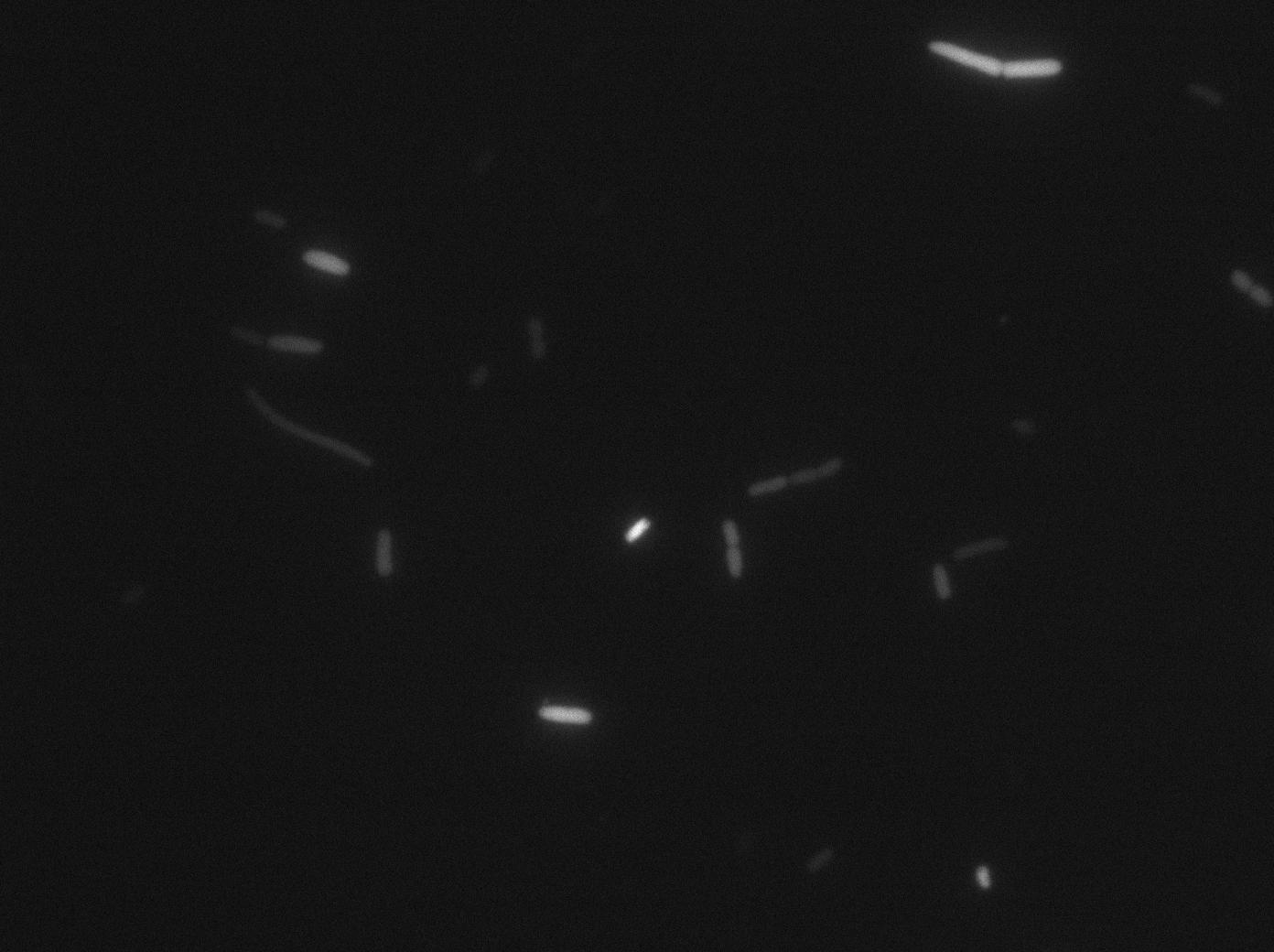
We characterized T7 autoloop in E.coli, when hosted in the plasmid pSB1C3 (
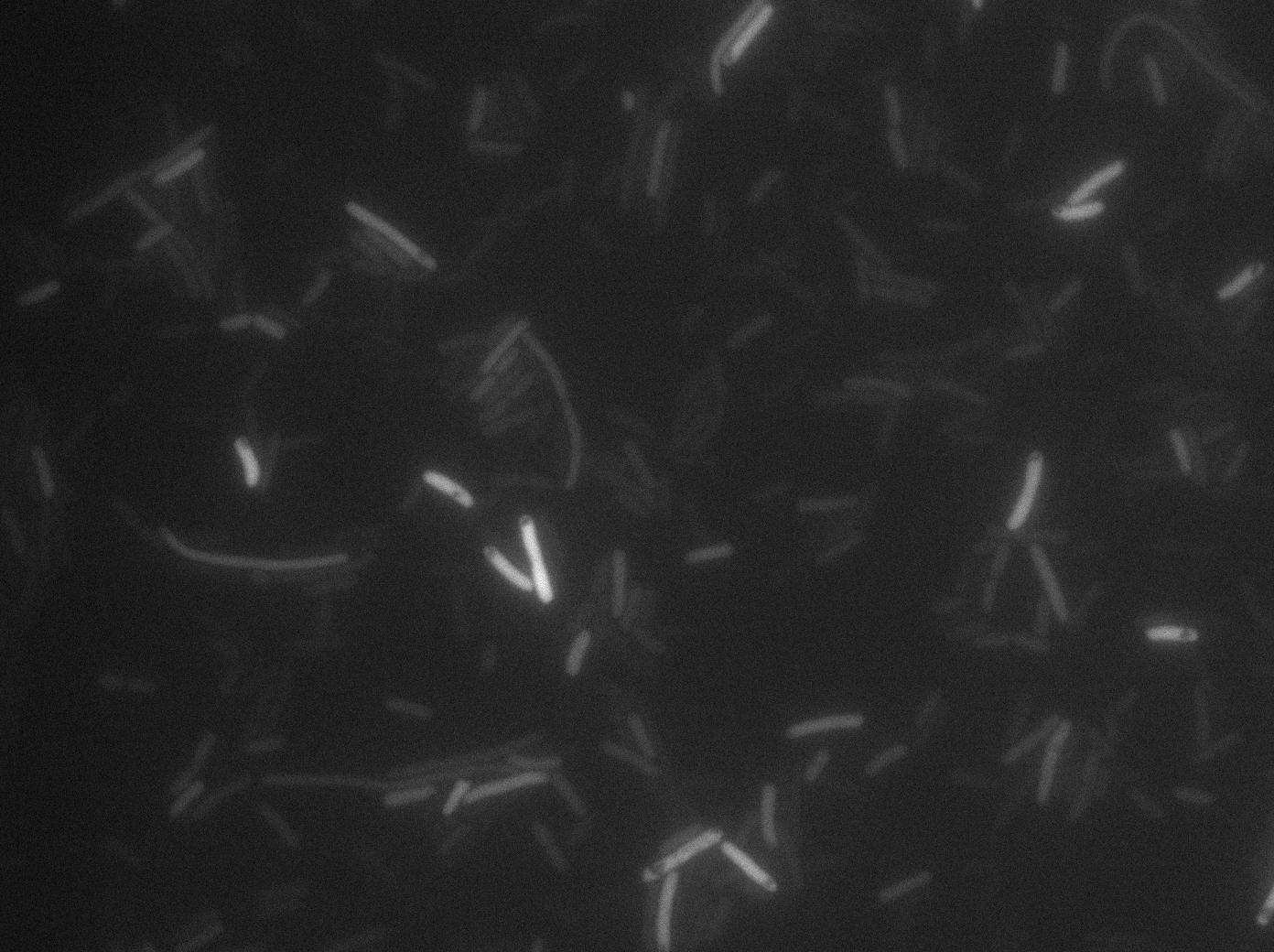
We found out there is a small leakage with the T7 emitter RFP. The cells in which RFP production is activated stop dividing and glow very strongly.
The first pictures show that the RFP construct is working efficiently since some cells are glowing with RFP fluorescence. This also shows the system is not as leaky as we expected. Indeed, the promoter regulating RFP expression is 'pVeg' which is a constitutive promoter. Finally, the RFP system is working very well because when leak occurs, cells glow very strongly.
These pictures show that the T7 GFP autoloop system is efficient since some cells are glowing with GFP fluorescence. Thus, we can conclude that the T7 polymerase is activated. Finally, we notice there is no difference between the GFP autoloop with and without terminator before the T7 promoter, we can interpret this due to this terminator is a B.subtilis terminator, moreover, we know that this E.coli strain has 4 terminators.
Perspectives
In order to get a better characterization of our systems and to tweak a wide range of parameters, we have already built a microfluidic device and are doing some preliminary experiments.
Afterwards, we want to clone the LacI expression system (<partinfo>BBa_K606054</partinfo>) into the emitter cell (<partinfo>BBa_K606039</partinfo>) in order to bring T7 under the control of LacI and thus permit IPTG induction.
Finally, we plan to insert these BioBricks into a replicative vector, like pHM3, or an integrative vector, like <partinfo>BBa_K090403</partinfo>, for B.subtilis in order to test the communication and the transfer of T7 polymerase through the nanotubes.
 "
"