Team:XMU-China/Project/Background
From 2011.igem.org
Contents |
Background
Quorum Sensing
Quorum sensing is a method of communication between bacteria that enables the coordination of group-based behavior based on population density. [3] It was first observed in Vibrio fischeri, a bioluminiscent bacterium that lives in the ocean. Bacteria that use quorum sensing constantly produce and secrete certain signaling molecules (called autoinducers or pheromones). These bacteria also have a receptor that can specifically detect the signaling molecule (inducer). When the inducer binds the receptor, it activates transcription of certain genes, including those for inducer synthesis. There is a low likelihood of a bacterium detecting its own secreted inducer. Thus, in order for gene transcription to be activated, the cell must encounter signaling molecules secreted by other cells in its environment. When only a few other bacteria of the same kind are in the vicinity, diffusion reduces the concentration of the inducer in the surrounding medium to almost zero, so the bacteria produce little inducer. However, as the population grows, the concentration of the inducer passes a threshold, causing more inducer to be synthesized. This forms a positive feedback loop, and the receptor becomes fully activated. Activation of the receptor induces the up-regulation of other specific genes, causing all of the cells to begin transcription at approximately the same time. This coordinated behavior of bacterial cells can be useful in a variety of situations. For instance, the bioluminescent luciferase produced by V. fischeri would not be visible if it were produced by a single cell. By using quorum sensing to limit the production of luciferase to situations when cell populations are large, V. fischeri cells are able to avoid wasting energy on the production of useless product.
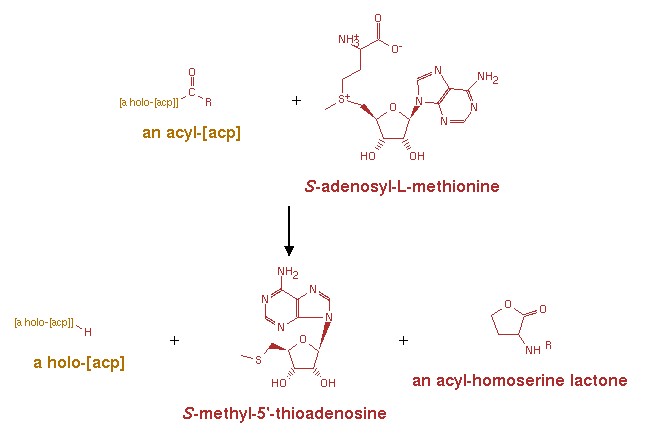
LuxI : Acyl-homoserine-lactone synthase
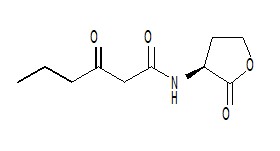
LuxI is required for the synthesis of OHHL (N-(3-oxohexanoyl)-L-homoserine lactone) also known as VAI or N-(beta-ketocaproyl)homoserine lactone or 3-oxo-N-(tetrahydro-2-oxo-3-furanyl)-hexanamide, an autoinducer molecule which binds to luxR and thus acts in bioluminescence regulation.[8]
Catalytic activity
An acyl-[acyl-carrier-protein] + S-adenosyl-L-methionine = [acyl-carrier-protein] + S-methyl-5'-thioadenosine + an N-acyl-L-homoserine lactone.[8]

LuxR: Transcriptional activator protein luxR
The function of LuxR homologues as quorum sensors is mediated by the binding of N-acyl-L-homoserine lactone (AHL) signal molecules to the N-terminal receptor site of the proteins.[1] It is a transcriptional activator of the bioluminescence operon. It binds to the AHL autoinducer.[7]
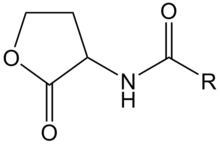
AHL: N-acyl-homoserine lactone
AHL is a kind of signaling molecule involved in bacterial quorum sensing.[2] In Vibrio fischeri, AHL binds to the protein product of the LuxR gene and activates it. The C-terminal domain of activated LuxR relieves the repression exerted by H-NS nucleoid proteins that bind to the promoters of LuxR, LuxI and the LuxCDABEG operon, as well as to A-T-rich stretches within that operon and other genomic regions. The product of LuxI catalyses the synthesis of AHL. Thus, AHL acts as an autoinducer. Transcription of the LuxCDABEG operon results in luminescence due to the expression of LuxA and LuxB, which form a protein known as a luciferase and the expression of LuxC, D, E, and G, which are involved in the synthesis of the luciferase's substrate, tetradecanal.[11]

IPTG: Isopropyl β-D-1-thiogalactopyranoside
This compound is used as a molecular mimic of allolactose, a lactose metabolite that triggers transcription of the lac operon. Many regulatory elements of the lac operon are used in inducible recombinant protein systems.[14]

iGEM-Team XMU-China has designed a series of circuits driven by The PLlac 0-1 promoter (BBa_R0011). IPTG was used as an inducer to activate these circuits.
toxin ccdB
Toxin ccdB is a component of toxin-antitoxin (TA) module, functioning in plasmid maintenance. Cell killing by CcdB is accompanied by filamentation, defects in chromosome and plasmid segregation, defects in cell division, formation of anucleate cells, decreased DNA synthesis and plasmid loss.
LacZalpha-ccdB
The LacZ alpha-CcdB fusion protein has retained both the CcdB killer activity and the ability to alpha-complement the truncated LacZ delta M15.
ccdB vs cell DEATH
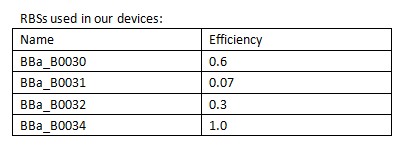
iGEM-Team XMU-China has designed and constructed a ccdB producer driven by promoter lux pR. We assumed that for circuit-regulated growth, the cell death rate is proportional to the intracellular concentration of the killer protein CcdB. For this reason, we controlled the expression level of the killer gene ccdB in three ways: (1) by using different RBSs (RBS1.0, RBS0.6, RBS0.3, RBS0.07); (2)by using different LuxR promoters(site-directed mutagenesis); (3) by using different LuxR(error-prone PCR).
RBS: Ribosome Binding Sites
A Ribosome Binding Site (RBS) is an RNA sequence found in mRNA to which ribosomes can bind and initiate translation. In order to control the expression of the killer protein ccdB, our team designed a series of bacteria population-control devices using RBSs with different strength. The cell growth and fluorescent curves corresponding to different RBSs illustrate that the bacteria population was successfully controlled at different cell densities.

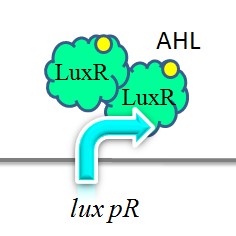
Lux pR
Promoter lux pR is activated by LuxR in concert with HSL (homoserine lactone). Two molecules of LuxR protein form a complex with two molecules of the signalling compound HSL. This complex binds to a palindromic site on the promoter, increasing the rate of transcription. This promoter is used in our “killer protein producer” to regulate the expression of the killer protein ccdB. iGEM-Team XMU-China has successfully strengthened the expression of lux pR by mutating its DNA sequence. Three mutants, IR-3, IR-5, IR-3/5, were obtained by site-directed mutagenesis using 3-step PCR method.

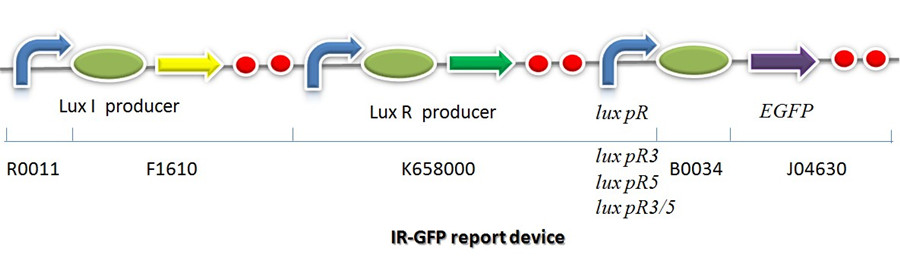
IR-GFP
IR-GFP is a series of report devices designed for testing the performance of lux R promoters before and after mutagenesis. These four IR-GFP report devices have been transformed into E.coli string BL21 separately. These devices produced greenish tint visible by naked eyes when induced by IPTG. We measured and compared their florescent intensities at steady state. As the only difference between the four devices is Lux R promoter, the efficiency of the four Lux R promoters could be defined.
reference
[1] Koch B, Liljefors T, Persson T , Nielsen J, Kjelleberg S, Givskov M. The LuxR receptor: the sites of interaction with quorum-sensing signals and inhibitors[J].Microbiology , 2005, 151 (11): 3589-3602. [2] Kumari A, Pasini P, Deo S. K., Flomenhoft D, Shashidhar S, Daunert S. Biosensing Systems for the Detection of Bacterial Quorum Signaling Molecules[J]. Analytical Chemistry,2005, 78 (22): 7603–7609. [3] Eberhard A, Burlingame A. L, Eberhard C, Kenyon G. L, Nealson K. H,Oppenheimer N. J. Structural identification of autoinducer of Photobacterium fischeri luciferase[J]. Biochemistry ,1981,20 (9): 2444–2449. [4] Gerdes K. Toxin-antitoxin modules may regulate synthesis of macromolecules during nutritional stress[J]. J. Bacteriol,2000,182 (3): 561–721.
[5]Philippe B,Martine C.Cell killing by the F plasmid CcdB protein involves poisoning of DNA-topoisomerase II complexes[J].Molecular Biology,1992,226(3):735-745.
[6]Devine J.H,Shadel G.S,Baldwin T.O. Identification of the operator of the lux regulon from the Vibrio fischeri strain ATCC7744. Proceedings of the National Academy of Sciences.1989.86(15):5688-5692.
[7]Miki T,Yoshioka K,Horiuchi T. Control of cell division by sex factor F in Escherichia coli. I. The 42.84-43.6 F segment couples cell division of the host bacteria with replication of plasmid DNA. Molecular Biology,1984,177(4):605-625.
[8] Hanzelka BL, Greenberg E. Quorum sensing in Vibrio fischeri: evidence that S-adenosyl methionine is the amino acid substrate for autoinducer synthesis[J]. Journal of Bacteriology, 1996, 178(17): 5291-5294.
[9] Urbanowski M, Lostroh C, Greenberg E. Reversible acyl-homoserine lactone binding to purified Vibrio fischeri LuxR protein[J]. Journal of Bacteriology, 2004, 186(3): 631-637.
[10] Hansen LH, Knudsen S, Sorensen SJ . The effect of the lacY gene on the induction of IPTG inducible promoters, studied in Escherichia coli and Pseudomonas fluorescens. Curr microbiol,1998, 36 (6): 341–345.
[11]Bernard P,Couturier M. Cell killing by the F plasmid CcdB protein involves poisoning of DNA-topoisomerase II complexes. Molecular Biology,1992,226:735-745.
[12]Semit M.H,Duin J. Secondary structure of the ribosome binding site determines translational efficiency: a quantitative analysis. Molecular Biology,1990,87(19):7668-7672.
[13] Baldwin T, Devine JH, Heckel, RC, Lin, JW, Shadel GS. The complete nucleotide sequence of the lux regulon of Vibrio fischeri and the luxABN region of Photobacterium leiognathi and the mechanism of control of bacterial bioluminescence[J]. Journal of Bioluminescence and Chemiluminescence, 1989, 4(1): 326-341.
[14] You L, Cox RS, Weiss R, Arnold FH. Programmed population control by cell-cell communication and regulated killing[J]. Nature, 2004, 428(6985): 868-871.
[15] http://en.wikipedia.org/wiki/N-Acyl_homoserine_lactone
[16] http://scholarworks.umass.edu/dissertations/AAI3397713/
[17] http://en.wikipedia.org/wiki/Toxin-antitoxin_system
[18] http://www.sciencedirect.com/science/article/pii/002228369290629X
[19] http://scholarworks.umass.edu/dissertations/AAI3397713/
[20] http://www.uniprot.org/uniprot/P12746
[21] http://www.uniprot.org/uniprot/P12747
[22] http://www.uniprot.org/uniprot/P62554
[23] http://www.nottingham.ac.uk/quorum/what2.htm
[24] http://www.humancyc.org/META/NEW-IMAGE?type=REACTION&object=2.3.1.184-RXN
[25] http://en.wikipedia.org/wiki/IPTG
[26] http://www.uniprot.org/uniprot/P62554
[27] http://en.wikipedia.org/wiki/Ribosomal_binding_site
 "
"