Team:Calgary/Project/Reporter
From 2011.igem.org
Rpgguardian (Talk | contribs) |
Emily Hicks (Talk | contribs) |
||
| Line 13: | Line 13: | ||
<p>Oilsands tailings ponds samples are difficult to work with for several different reasons. First of all, tailings ponds samples are murky, and can have greatly varying compositions from pond to pond. It would be difficult to obtain accurate colorimetric data from these samples, and it would be similarly difficult to readily observe fluorescence. Because an electrochemical response does not rely on being able to see anything, Team Calgary determined the electrochemical reporter to be ideal for this purpose.</p> | <p>Oilsands tailings ponds samples are difficult to work with for several different reasons. First of all, tailings ponds samples are murky, and can have greatly varying compositions from pond to pond. It would be difficult to obtain accurate colorimetric data from these samples, and it would be similarly difficult to readily observe fluorescence. Because an electrochemical response does not rely on being able to see anything, Team Calgary determined the electrochemical reporter to be ideal for this purpose.</p> | ||
| - | <p>Electrochemical reporters offer several advantages over | + | <p>Electrochemical reporters offer several advantages over colorimetric and flourometric reporters: the signal is robust enough to not be altered by the presence of pollutants or sample turbidity do not affect the signal. Every molecule has a different oxidation potential - because we know the exact oxidation potential for our analyte, we can easily tell the difference between our desired signal and background noise. Another advantage of an electrochemical response is that the data is immediately available in digital form. This allows for fast graphical interpretation of the data as well as maximum information collection from the data. </p> |
Revision as of 00:33, 29 September 2011









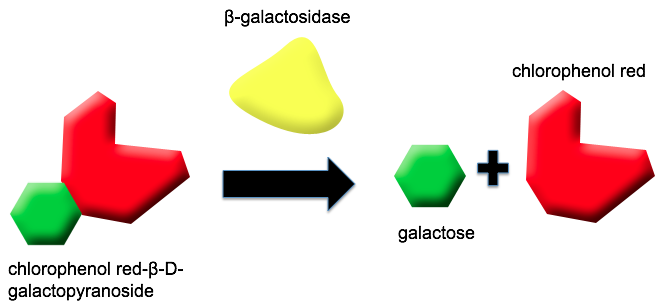
Reporter

What is a Reporter?
Being able to detect a particle in laboratory conditions can vary drastically from conditions in situ. After the presence of naphthenic acids is confirmed by our promoter, we will require a reporter system for our bacteria to report accurately and decisively. Over the course of the project, Team Calgary considered three different reporter systems - colorimetric, fluorescent, and electrochemical.
Why Electrochemical?
Oilsands tailings ponds samples are difficult to work with for several different reasons. First of all, tailings ponds samples are murky, and can have greatly varying compositions from pond to pond. It would be difficult to obtain accurate colorimetric data from these samples, and it would be similarly difficult to readily observe fluorescence. Because an electrochemical response does not rely on being able to see anything, Team Calgary determined the electrochemical reporter to be ideal for this purpose.
Electrochemical reporters offer several advantages over colorimetric and flourometric reporters: the signal is robust enough to not be altered by the presence of pollutants or sample turbidity do not affect the signal. Every molecule has a different oxidation potential - because we know the exact oxidation potential for our analyte, we can easily tell the difference between our desired signal and background noise. Another advantage of an electrochemical response is that the data is immediately available in digital form. This allows for fast graphical interpretation of the data as well as maximum information collection from the data.
A Novel Application for the lacZ Gene
Building our System
One of the main reasons we selected a electrochemical response for our system was that the response can be detected and processed digitally, without the need for qualitative observations. Because all molecules have a different oxidation potential, we can easily detect the presence or absence of an analyte with a known oxidation potential.
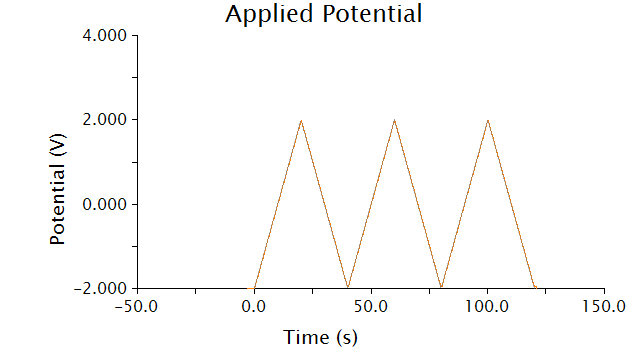
The voltammetric technique we used to detect chlorophenol red was cyclic voltammetry - the input voltage is shown below.
As voltage slowly increases and decreases, each species present in solution will undergo a redox reaction at its respective oxidation potential. Because we know the oxidation potential of our analyte, chlorophenol red (CPR), we can ignore any background noise or pollution using this method.
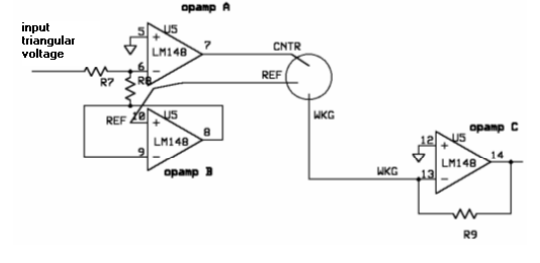
A three-electrode setup is required to perform cyclic voltammetry. The electrical current passes between the working and counter electrodes - and the potential difference between the third electrode and counter electrode is controlled. This setup allows us to take voltage and current readings that do not interfere with each other.
In order to perform cyclic voltammetry, specialized equipment is usually required. Because such equipment is usually unavailable in biology labs, we built our own potentiostat - a piece of equipment that outputs a known voltage and measures a corresponding current across two electrodes. Although we used a commercially available potentiostat to conduct our experiments, we decided to build our own to demonstrate that a cheap, reliable set-up is feasible.
For preliminary data, click here.For characterization data, click here.

 "
"