Team:Cambridge/Project/Microscopy
From 2011.igem.org
(→Reflectin Thin Films) |
|||
| Line 44: | Line 44: | ||
File:BSAcontrolfilm1.jpg| ''Bovine Serum Albumin makes a dull, striated thin film'' | File:BSAcontrolfilm1.jpg| ''Bovine Serum Albumin makes a dull, striated thin film'' | ||
</gallery> | </gallery> | ||
| + | |||
| + | [[File:Cam Multilayer drop 1.jpg | thumb | 600px | center |A light micrograph of...]] | ||
| + | |||
| + | [[File:Cam Crazy multilayer single AP 2k spin2nd.jpg | thumb | 600px | center |A light micrograph of...]] | ||
| + | |||
| + | [[File:Cam crazy multilayer2.jpg| thumb | 600px | center |A light micrograph of...]] | ||
<br style='clear:both' /> | <br style='clear:both' /> | ||
{{Template:Team:Cambridge/CAM_2011_TEMPLATE_FOOT}} | {{Template:Team:Cambridge/CAM_2011_TEMPLATE_FOOT}} | ||
Revision as of 12:37, 20 September 2011
Contents |
Microscopy
Preliminary observations
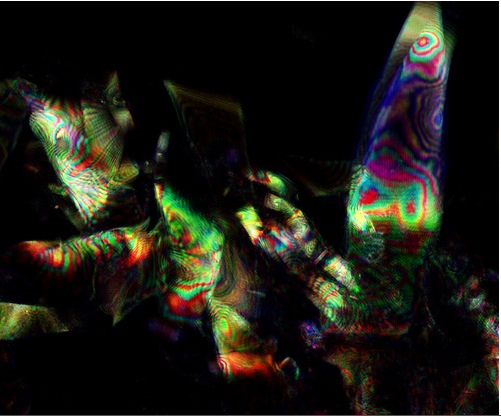
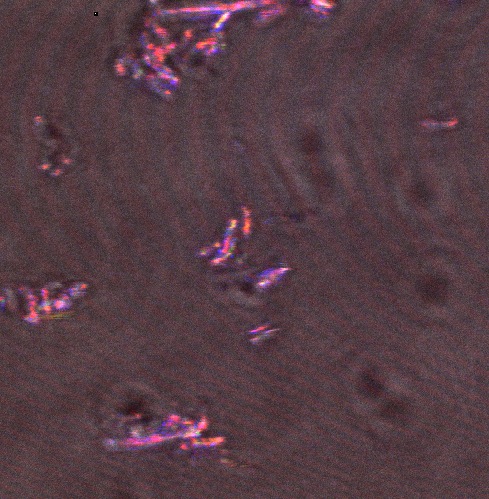
In order to get a real sense of what we were looking to achieve in our project, we felt that it was important to make some observations of native squid reflectin in vivo. We therefore obtained several specimens of Loligo opalescens and Loligo vulgaris squid from a local seafood restaurant and an online fishing bait store for dissection. We chose these species because the whole family of loliginid squid has been identified to contain reflectin, and these particular species were the only members of the family available to us. We used a confocal microscope to observe iridescent behaviour in eye and mantle tissue, by the following protocol. The stunning images produced provided a very useful reference to help us to identify what recombinant (well folded) reflectin could look like in E. coli, and definitely enthused the team to obtain bactiridescence!
Squid Tissues
We set the microscope to collect light reflected from the sample (emission and collection wavelengths overlap) as we were searching for iridescence. We'd like to thank Paul Grant who optimised the settings on the microscope. We then overlaid the images ourselves to produce the animated gif on the right.
We are very grateful to Fernan Federici who helped us, taking the image below using the 405nm, 488nm, 633nm laser beams and with the pinhole opened to a wider aperture.
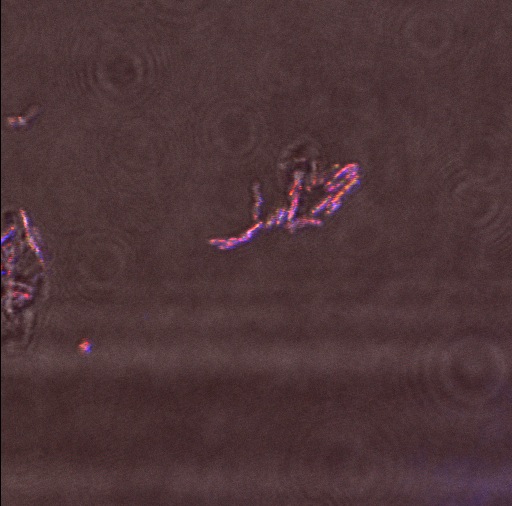
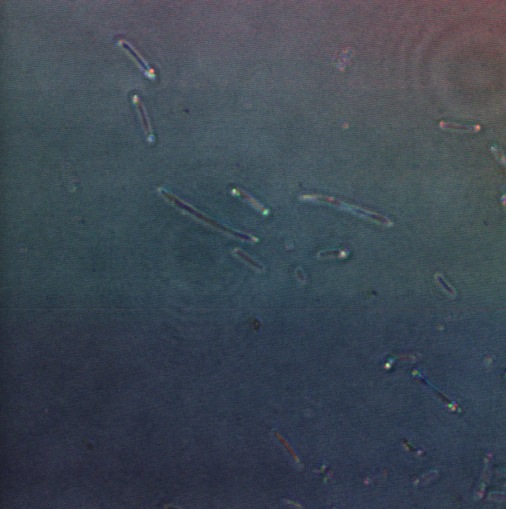
We used the enzyme trypsin to isolate cells from the mantle of the squid as this frees them from the extracellular matrix. Again we used the confocal microscope in a configuration that detects reflected light.
Reflectin Expressing Cells
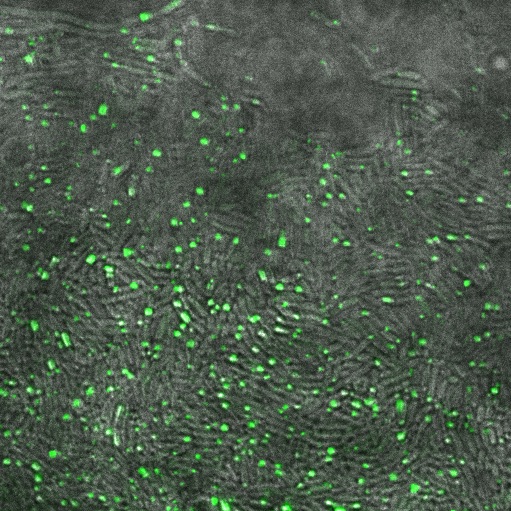
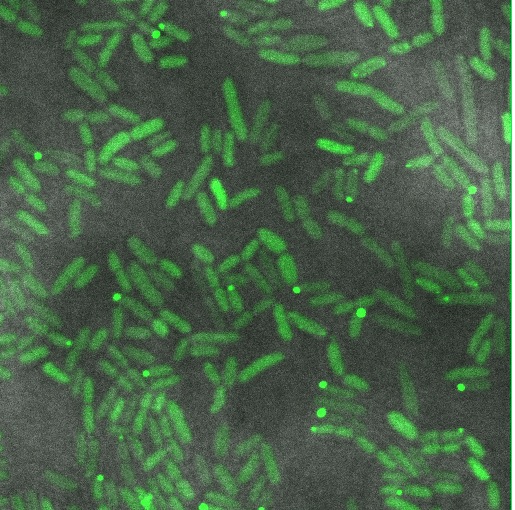
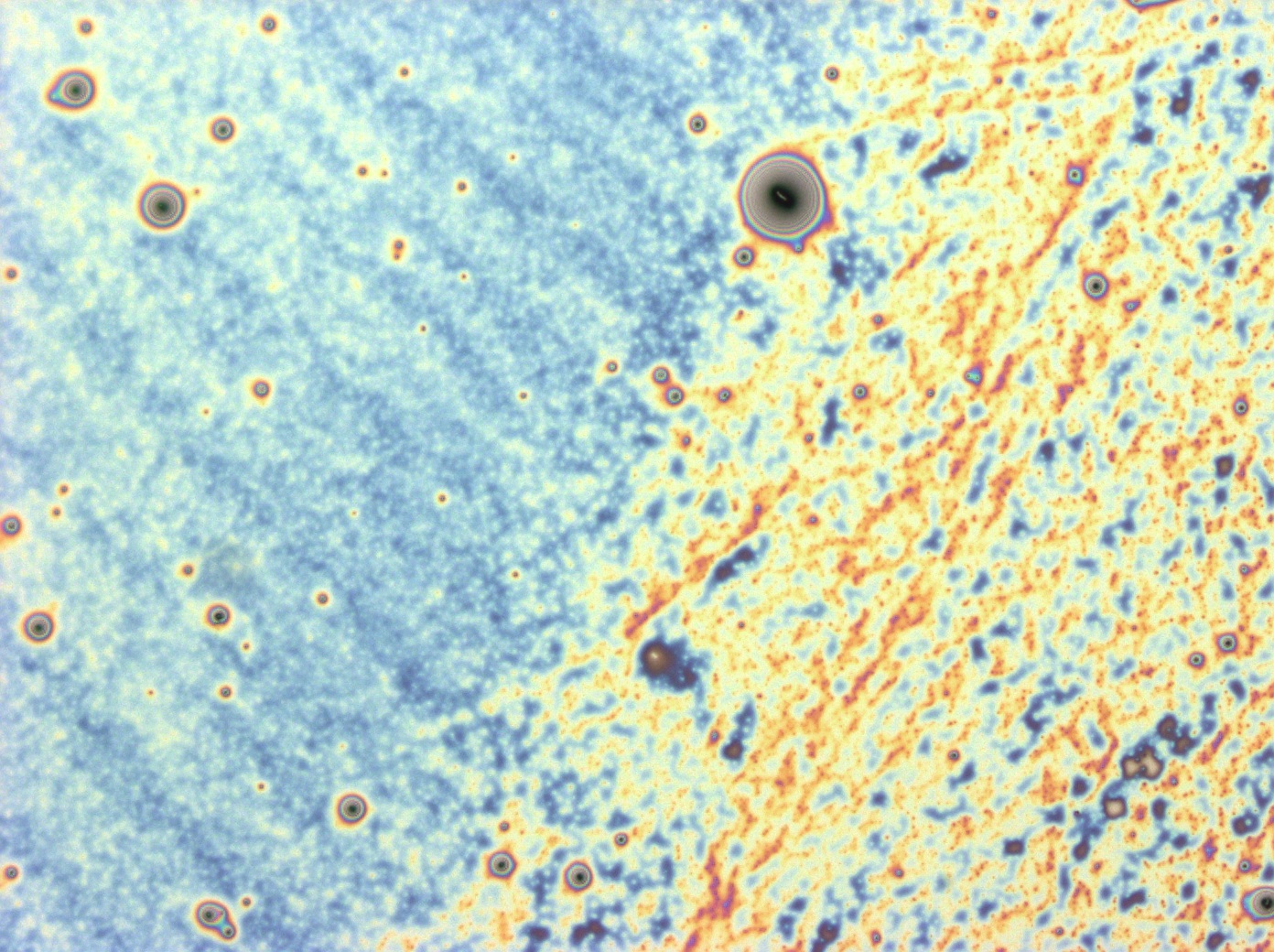
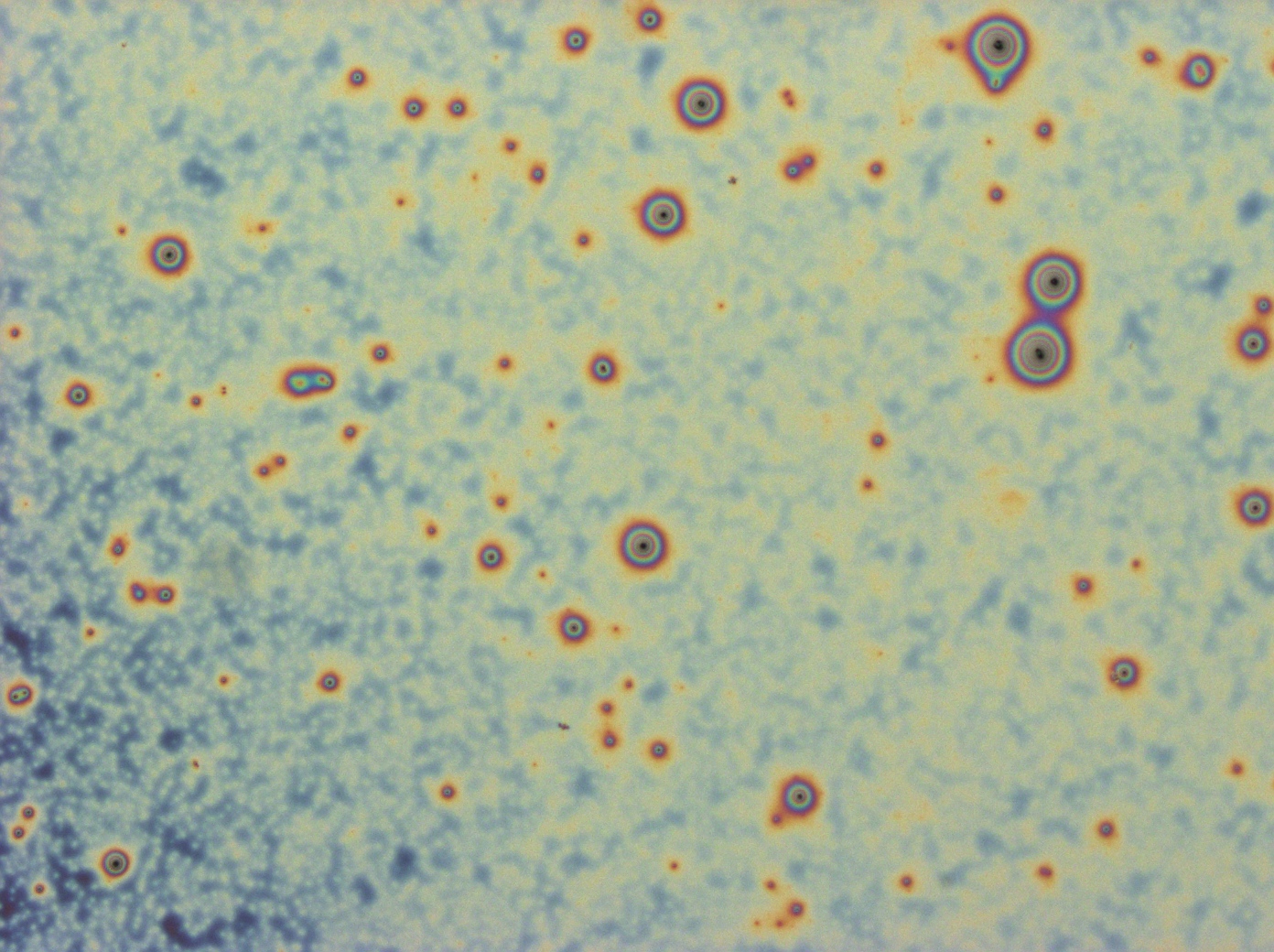
These cells are producing a reflectin A1-GFP fusion protein and display display fluorescent inclusion bodies. Suggesting expression of the protein is too high for the protein to fold properly.
We fused a TorA export sequence to a reflectin A1-green fluorescent protein fusion in an attempt to export reflectin to the periplasm of E coli. Unfortunately, even on a low copy plasmid, it appears that the export tag has failed or we have saturated the export pathway and a backlog has occured. This means all cells have a detectable amount of GFP in their cytoplasm and fluorescent inclusion bodies in several of the transformed cells.
The following confocal micrographs show control bacteria, transformed but uninduced bacteria and bacteria containing our construct and induced to produce reflectin A1. Interestingly, all cells display some reflectance, and perhaps surprsingly the induced cells show the least overall reflectanct. The induced cells display punctate dots of reflectance, which suggests inclusion bodies have been formed. However, despite uniform imaging settings across the data set, there is a differing amount of reflectance from the medium the cells are growing on. Further work we would like to carry out would involve producing slides of the same bacteria but using gelatin and PBS to minimise the background reflectance, compare reflectin and non-reflectin inclusion bodies under various imaging techniques and make use of a spectrometer to gather spectral data on the reflectin expressing bacteria, as the limitations of reflected light confocal microscopy - namely background reflectance, is highlighted by these images.
Reflectin Thin Films
 "
"