|
|
| (8 intermediate revisions not shown) |
| Line 1: |
Line 1: |
| - | {{:Team:EPF-Lausanne/Templates/TetRtemplate|title=Intro}} | + | {{:Team:EPF-Lausanne/Templates/TetRtemplate|title=''In-Vitro'' Characterization}} |
| | | | |
| - | After having considered several potential transcription factor candidates, the choice was made to use TetR for our transcription factor development pipeline. TetR has several advantages:
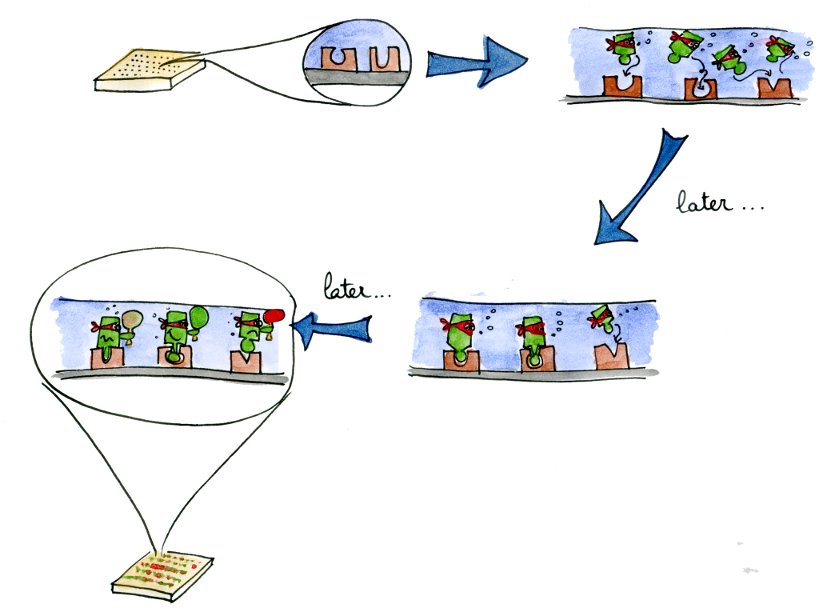
| + | When we generate new libraries of transcription factors using site-directed mutagenesis, we need to have a precise and high-throughput characterization method. These two requirements are met by the microfluidics MITOMI device that we used. |
| - | * It exhibits a strong affinity to its consensus sequence
| + | |
| - | * It has already been well characterized in the literature – especially the region of the protein involved in promoter recognition has already been described. TetR mutants showing an altered specificity have already been reported – showing that our idea is feasible
| + | |
| - | * TetR is one of the most used transcription factors used in synthetic biology – expanding its properties would be of great interest
| + | |
| - | * It can be easily repressed by adding anhydrotetracycline (ATC) – a cheap molecule
| + | |
| - |
| + | |
| | | | |
| - | == Background Information about TetR ==
| + | Characterizing a lot of different mutants will allow us to get a better understanding of the mutations influencing the specificity and affinity of a transcription factor. With these parameters in hand, we will be able to fine-tune the characteristics of the newly produced transcription factors. |
| | | | |
| - | Widespread among bacteria, TetR is a '''repressor''' that regulates enzymes essential for resistance against the antibiotic tetracycline. These enzymes are encoded on the tet operon (TetO), which is repressed by TetR in normal conditions. When tetracycline is present, the antibiotic molecule binds to TetR and inactivates it, thus allowing the expression of TetO.
| + | The new transcription factors mutants characterized in vitro then need to be tested ''in vivo''. This last step is explained in the " ''in vivo'' characterization section". |
| - | | + | |
| - | TetR forms a dimer, each part of the dimer being involved in DNA binding. Consequently, the recognition sequence of TetR is symmetrical (the two boxes on the image below), with a base pair separating the two sub-sequences. [http://www.nature.com/doifinder/10.1038/73324 Orth et al, 2000]
| + | |
| - | The whole promoter, comprising this recognition sequence, is called Ptet.
| + | |
| - | | + | |
| - | [[File:EPFL_TetR_consensus.jpg]]
| + | |
| - | | + | |
| - | | + | |
| - | The binding of each monomer to the recognition sequence has been studied thoroughly; we know which amino acid interacts to which nucleotide, as can be seen in the figure below. The amino acids directly involved in binding to the DNA are found in between position 26 and 48 of each monomer. [http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1197418/?tool=pubmed Ramos et al, 2005]
| + | |
| - | | + | |
| - | [[File:EPFL_TetR_contact_map.jpg|500px]]
| + | |
| - | | + | |
| - | | + | |
| - | The impact of one or several amino acids changes on the binding strength and specificity is less known. Amino acids in direct proximity to or the residues binding DNA are probably crucial - but more distant amino acids could also have an influence due to allosteric effects. Some TetR mutants that have an altered recognition sequence have been characterized in the literature.
| + | |
| - | | + | |
| - | The effects of the mutations V36F, E37A, P39K and Y42F were described in [http://www.sciencedirect.com/science/article/pii/S0378111907004623 Krueger et al, 2007].
| + | |
| - | [http://www.sciencedirect.com/science/article/pii/S0022283697915400 Helbl et al, 1998] states other interesting mutants: the P39Q mutation was shown to exhibit a new recognition specificity for the tetO-4C operator and the E37A Y42M P39Q said to have the highest affinity for tetO-4C.
| + | |
| | | | |
| | + | [[File:EPFL-Solange-MITOMI.jpg|700px]] |
| | | | |
| | {{:Team:EPF-Lausanne/Templates/Footer}} | | {{:Team:EPF-Lausanne/Templates/Footer}} |
In-Vitro Characterization
In vitro Main | Why TetR? | Mutant TetRs | MITOMI Data | In-vivo & In-vitro outline
When we generate new libraries of transcription factors using site-directed mutagenesis, we need to have a precise and high-throughput characterization method. These two requirements are met by the microfluidics MITOMI device that we used.
Characterizing a lot of different mutants will allow us to get a better understanding of the mutations influencing the specificity and affinity of a transcription factor. With these parameters in hand, we will be able to fine-tune the characteristics of the newly produced transcription factors.
The new transcription factors mutants characterized in vitro then need to be tested in vivo. This last step is explained in the " in vivo characterization section".
 "
"
