Team:Cambridge/Project/In Vitro
From 2011.igem.org
| Line 1: | Line 1: | ||
{{Template:Team:Cambridge/CAM_2011_TEMPLATE_HEAD}} | {{Template:Team:Cambridge/CAM_2011_TEMPLATE_HEAD}} | ||
| - | = | + | =In Vitro – Isolating Reflectin and Making Thin Films= |
| + | Previous in vitro investigations of reflectin showed that it was possible to use a method called flow coating to deposit a thin layer of reflectin onto a silicon substrate which would then demonstrate structural colour. | ||
| - | + | We wanted to investigate using a different coating method – spin coating – and to try out different methods for protein purification. A HIS-tagged variant of the protein was overexpressed and purified using a HIS trap. Thin films were made by precipitating with either acetone or ethanol and resuspending in HFIP before either spin coating or flow coating onto a silicon wafer. | |
| + | |||
| + | We also investigated an inclusion body prep, as the protein we found that the protein formed inclusion bodies upon over-expression. | ||
| + | |||
| + | By varying our purification and coating methods, we were able to create vibrant thin films. | ||
[[File:CAM_invitro_flow.png| centre | 300px]] | [[File:CAM_invitro_flow.png| centre | 300px]] | ||
| - | ==Tagging | + | ==HIS-Tagging Reflectin== |
| + | |||
| + | The reflectin genes were HIS-tagged by incorporating the sequence of part [http://partsregistry.org/wiki/index.php?title=Part:BBa_K128005 BBa_K128005] into the primers used to clone the gene. HIS tagging was chosen because it allows proteins to be purified with relatively simple apparatus, a plastic column, to which a metal containing resin is added traps the protein when cell lysate is added. | ||
| + | |||
| + | ==Over-Expression== | ||
| - | + | Reflectin was placed on a high copy plasmid ([http://partsregistry.org/Part:pSB1A2 PSB1A2]) under an arabinose inducible promoter ([http://partsregistry.org/Part:BBa_I0500 pBad]) in order to express reflectin at a high level. As well as a negative control, we used an sfGFP-reflectin fusion as a control for reflecitn production. | |
| - | + | In the non-GFP cells we found that there were brightly lit points at the ends of nearly every cell, which were not present in the negative control. In the GFP fusion cells, these spots glowed strongly, while the rest of the cell was relatively dark. This indicated that reflectin produces inclusion bodies when expressed at high level. | |
| - | We | + | <html> |
| + | <p style='color:red;font-style:italic;'>We really need the images here!</p> | ||
| + | </html> | ||
| - | == | + | ==Extraction & Purification== |
A number of protein purification protocols were used to isolate the reflectin-containing inclusion bodies from transformed E. coli. | A number of protein purification protocols were used to isolate the reflectin-containing inclusion bodies from transformed E. coli. | ||
| - | The Norgen Proteospin inclusion body prep. kit was experimented-with as a commercial alternative to the ethanol/acetone precipitations used previously. The kit is simple to use, but does require access to an ultracentrifuge capable of spinning up to 25ml of fluid at 27,000g. The latter stages of the protocol require numerous uses of a | + | The Norgen Proteospin inclusion body prep. kit was experimented-with as a commercial alternative to the ethanol/acetone precipitations used previously. The kit is simple to use, but does require access to an ultracentrifuge capable of spinning up to 25ml of fluid at 27,000g. The latter stages of the protocol require numerous uses of a benchtop centrifuge. It was found that the spinning forces specified in the protocol for these steps weren't capable of moving fluid through the necessary columns and higher forces were needed. On analysing the end-product of the purification with SDS-PAGE approximately 20 discrete bands were observed from 10-200 kda with 2 bold bands, one of which corresponded to the size of reflectin. |
| - | benchtop centrifuge. It was found that the spinning forces specified in the protocol for these steps weren't capable of moving fluid through the necessary columns and higher forces were needed. On analysing the end-product of the purification with SDS-PAGE approximately 20 discrete bands were observed from 10-200 kda with 2 bold bands, one of which corresponded to the size of reflectin. | + | |
{{Template:Team:Cambridge/CAM_2011_TEMPLATE_FOOT}} | {{Template:Team:Cambridge/CAM_2011_TEMPLATE_FOOT}} | ||
Revision as of 11:37, 19 September 2011
Contents |
In Vitro – Isolating Reflectin and Making Thin Films
Previous in vitro investigations of reflectin showed that it was possible to use a method called flow coating to deposit a thin layer of reflectin onto a silicon substrate which would then demonstrate structural colour.
We wanted to investigate using a different coating method – spin coating – and to try out different methods for protein purification. A HIS-tagged variant of the protein was overexpressed and purified using a HIS trap. Thin films were made by precipitating with either acetone or ethanol and resuspending in HFIP before either spin coating or flow coating onto a silicon wafer.
We also investigated an inclusion body prep, as the protein we found that the protein formed inclusion bodies upon over-expression.
By varying our purification and coating methods, we were able to create vibrant thin films.
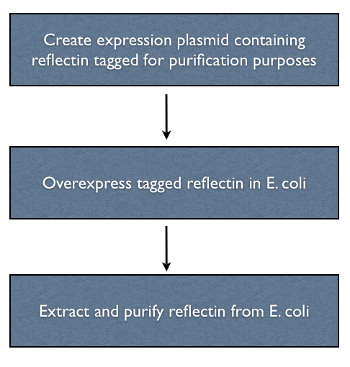
HIS-Tagging Reflectin
The reflectin genes were HIS-tagged by incorporating the sequence of part BBa_K128005 into the primers used to clone the gene. HIS tagging was chosen because it allows proteins to be purified with relatively simple apparatus, a plastic column, to which a metal containing resin is added traps the protein when cell lysate is added.
Over-Expression
Reflectin was placed on a high copy plasmid (PSB1A2) under an arabinose inducible promoter (pBad) in order to express reflectin at a high level. As well as a negative control, we used an sfGFP-reflectin fusion as a control for reflecitn production.
In the non-GFP cells we found that there were brightly lit points at the ends of nearly every cell, which were not present in the negative control. In the GFP fusion cells, these spots glowed strongly, while the rest of the cell was relatively dark. This indicated that reflectin produces inclusion bodies when expressed at high level.
We really need the images here!
Extraction & Purification
A number of protein purification protocols were used to isolate the reflectin-containing inclusion bodies from transformed E. coli.
The Norgen Proteospin inclusion body prep. kit was experimented-with as a commercial alternative to the ethanol/acetone precipitations used previously. The kit is simple to use, but does require access to an ultracentrifuge capable of spinning up to 25ml of fluid at 27,000g. The latter stages of the protocol require numerous uses of a benchtop centrifuge. It was found that the spinning forces specified in the protocol for these steps weren't capable of moving fluid through the necessary columns and higher forces were needed. On analysing the end-product of the purification with SDS-PAGE approximately 20 discrete bands were observed from 10-200 kda with 2 bold bands, one of which corresponded to the size of reflectin.
 "
"