Team:Freiburg/Notebook/14 July
From 2011.igem.org
(Difference between revisions)
(Created page with "==<span style="color:green;">green light receptor</span>== ===NAME OF YOUR EXPERIMENT=== '''Investigators:NAME''' ==<span style="color:blue;">blue light receptor</span>== =...") |
|||
| (23 intermediate revisions not shown) | |||
| Line 1: | Line 1: | ||
| - | ==< | + | {{:Team:Freiburg/Templates/header}} |
| + | <html> | ||
| + | <div id="notebook-page-header"> | ||
| + | <div id="notebook-back" width="100px" > | ||
| + | <a href="https://2011.igem.org/Team:Freiburg/Notebook/13_July">Previous entry</a> | ||
| + | </div> | ||
| + | <div id="notebook-title"> | ||
| + | <a href="https://2011.igem.org/Team:Freiburg/Notebook"> 14 July </a> | ||
| + | </div> | ||
| + | <div id="notebook-next"> | ||
| + | <a href="https://2011.igem.org/Team:Freiburg/Notebook/15_July">Next entry</a> | ||
| + | </div> | ||
| + | </div> | ||
| + | </html> | ||
| - | === | + | ==<span style="color:red;">red light receptor</span>== |
| - | + | ===3A-assembly: Digest=== | |
| + | '''Investigators: Jakob''' | ||
| + | 3A-assembly of S22 (ho1)+S21a (terminator) and S23 (pcyA)+S21a (terminator) | ||
| - | |||
| - | + | '''Digestion''' | |
| - | |||
| + | {| style="border-spacing:0;" | ||
| + | | style="border-top:0.0139in solid #808080;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| Name:Jakob | ||
| + | | style="border:0.0139in solid #808080;padding:0.0194in;"| Date: 14.07.2011 | ||
| + | |- | ||
| + | | colspan="2" style="border:0.0139in solid #808080;padding:0.0194in;"| Project Name: 3A-assembly of pcyA+ terminator, ho1+terminator | ||
| + | |} | ||
| + | | ||
| - | |||
| - | + | Procedure | |
| - | + | | |
| + | 1. add H2O (16μl-DNA ) | ||
| + | 2. 2μl NEB4 buffer | ||
| + | 3. 2 μl 10x BSA (used 1:10 diluted ) | ||
| + | |||
| + | 4. DNA (500 ng) (Vector:Insert ratio 1:3 in following Ligation) | ||
| + | |||
| + | 5. 1 μl restriction enzymes | ||
| + | |||
| + | 6. heat for 1-2 hours 37°C (6 hours if time) | ||
| + | |||
| + | 7. heat for 20 minutes 80°C (inactivation of enzymes) | ||
| + | |||
| + | 8. keep at 4°C if you cannot continue | ||
| + | |||
| + | | ||
| + | |||
| + | Measured DNA-concentration with Nanodrop to calculate the volume of DNA to do the digestion: | ||
| + | |||
| + | |||
| + | {| style="border-spacing:0;" | ||
| + | | style="border-top:0.0139in solid #808080;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| Sample Name | ||
| + | | style="border:0.0139in solid #808080;padding:0.0194in;"| DNA concentration (μg/μl) | ||
| + | |||
| + | |- | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| Vector pSB1T3 | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:0.0139in solid #808080;padding:0.0194in;"| 20 | ||
| + | |||
| + | |- | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| terminator | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:0.0139in solid #808080;padding:0.0194in;"| 38.8 | ||
| + | |||
| + | |- | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| ho1 | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:0.0139in solid #808080;padding:0.0194in;"| 176 | ||
| + | |||
| + | |- | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| pcyA | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:0.0139in solid #808080;padding:0.0194in;"| 173 | ||
| + | |||
| + | |} | ||
| + | | ||
| + | |||
| + | Restriction enzymes you need to cut the vector, insert1 and insert 2: | ||
| + | |||
| + | |||
| + | {| style="border-spacing:0;" | ||
| + | | style="border-top:0.0139in solid #808080;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| '''Components''' | ||
| + | | style="border-top:0.0139in solid #808080;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| <center>'''Vector (μl)'''</center> | ||
| + | | colspan="2" style="border:0.0139in solid #808080;padding:0.0194in;"| <center>'''Insert1 and 2 (μl)'''</center> | ||
| + | |||
| + | |- | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| DNA (μl) | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| 2 | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| Ho1 2.5 | ||
| + | |||
| + | pcyA 2.5 | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:0.0139in solid #808080;padding:0.0194in;"| Terminator 5.5 | ||
| + | |||
| + | terminator 5.5 | ||
| + | |||
| + | |- | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| BSA (10x) (2μl) | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:0.0139in solid #808080;padding:0.0194in;"| | ||
| + | |||
| + | |- | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| NEB4 Buffer (2μl) | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:0.0139in solid #808080;padding:0.0194in;"| | ||
| + | |||
| + | |- | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| Enzyme 1 (1μl) | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| EcoRI | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| EcoRI | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:0.0139in solid #808080;padding:0.0194in;"| XbaI | ||
| + | |||
| + | |- | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| Enzyme 2 (1μl) | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| PstI | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| SpeI | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:0.0139in solid #808080;padding:0.0194in;"| PstI | ||
| + | |||
| + | |- | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| H2O (14 μl- DNA) | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:0.0139in solid #808080;padding:0.0194in;"| | ||
| + | |||
| + | |- | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| In total 20 μl | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:0.0139in solid #808080;padding:0.0194in;"| | ||
| + | |||
| + | |} | ||
| + | | ||
| + | |||
| + | ===3A-assembly: Ligation and transformation=== | ||
| + | |||
| + | '''Investigators: Julia''' | ||
| + | |||
| + | Ligation of S22 (ho1)+S21a (terminator) and S23 (pcyA)+S21a (terminator) in vector pSB1T3. | ||
| + | |||
| + | L23: Ligation of S23+S21a in pSB1T3 | ||
| + | L22: Ligation of S22+S21a in pSB1T3 | ||
| + | '''Ligation''' | ||
| + | |||
| + | |||
| + | {| style="border-spacing:0;" | ||
| + | | style="border-top:0.0139in solid #808080;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| Name:Julia | ||
| + | | style="border:0.0139in solid #808080;padding:0.0194in;"| Date:14.07 | ||
| + | |||
| + | |- | ||
| + | | colspan="2" style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:0.0139in solid #808080;padding:0.0194in;"| Continue from Date14.07 Name | ||
| + | |||
| + | Experiment 3A-assembly | ||
| + | |||
| + | |- | ||
| + | | colspan="2" style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:0.0139in solid #808080;padding:0.0194in;"| Project Name:3A-assembly of pcyA+ terminator, ho1+terminator | ||
| + | |||
| + | |} | ||
| + | | ||
| + | |||
| + | |||
| + | '''Procedure''' | ||
| + | |||
| + | total volume 20 μl | ||
| + | |||
| + | |||
| + | 1. add H2O(17 μl -X-Y-Z) | ||
| + | |||
| + | 2. add 2 μl Ligase Buffer 10x | ||
| + | |||
| + | 3. add Insert 1, Insert 2 (when proceeding from 3A digestion use 2 μl of each) | ||
| + | |||
| + | 4. add Vector (20ng needed. When proceeding from 3A digestion use 2 μl) | ||
| + | |||
| + | 5. Add 1 μl T4-DNA Ligase | ||
| + | |||
| + | 6. Incubate 10-30 min at room temperature | ||
| + | |||
| + | 7. heat for 20 minutes at 80°C | ||
| + | |||
| + | 8. store at -20°C or directly proceed to transformation | ||
| + | |||
| + | | ||
| + | |||
| + | |||
| + | {| style="border-spacing:0;" | ||
| + | | style="border-top:0.0139in solid #808080;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| | ||
| + | | style="border-top:0.0139in solid #808080;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| Name of part | ||
| + | | style="border-top:0.0139in solid #808080;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| Ratio Insert:Vector | ||
| + | |||
| + | 1:1 | ||
| + | | style="border:0.0139in solid #808080;padding:0.0194in;"| Volume (μl) | ||
| + | |||
| + | |- | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| X insert 1 | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| ho1/ pcyA | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:0.0139in solid #808080;padding:0.0194in;"| 2 | ||
| + | |||
| + | |- | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| Y insert 2 | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| terminator | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:0.0139in solid #808080;padding:0.0194in;"| 2 | ||
| + | |||
| + | |- | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| Z vector | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| pSB1C3 | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:0.0139in solid #808080;padding:0.0194in;"| 2.5 | ||
| + | |||
| + | |- | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| H2O | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:none;padding:0.0194in;"| | ||
| + | | style="border-top:none;border-bottom:0.0139in solid #808080;border-left:0.0139in solid #808080;border-right:0.0139in solid #808080;padding:0.0194in;"| 11 | ||
| + | |||
| + | |} | ||
| + | |||
| + | |||
| + | Competent neb10 strain was transformed with 2μl ligation reaction and plated out. | ||
| + | |||
| + | ===PCR=== | ||
| + | |||
| + | '''Investigators: Julia''' | ||
| + | |||
| + | PCR of the red light receptor, cph8. | ||
| + | |||
| + | Primer: | ||
| + | |||
| + | {| style="border-spacing:0;" | ||
| + | | style="border-top:0.0007in solid #000000;border-bottom:0.0007in solid #000000;border-left:0.0007in solid #000000;border-right:none;padding:0.0382in;"| P10 | ||
| + | | style="border:0.0007in solid #000000;padding:0.0382in;"| atatgaattcgcggccgcttctagATGGCCACCACCGTAC | ||
| + | |||
| + | |- | ||
| + | | style="border-top:none;border-bottom:0.0007in solid #000000;border-left:0.0007in solid #000000;border-right:none;padding:0.0382in;"| P11 | ||
| + | | style="border-top:none;border-bottom:0.0007in solid #000000;border-left:0.0007in solid #000000;border-right:0.0007in solid #000000;padding:0.0382in;"| atatatgcatctgcagcggccgctactagtaTTACCCTTCTTTTGTCaTGCC | ||
| + | |||
| + | |} | ||
| + | |||
| + | |||
| + | |||
| + | DNA template: | ||
| + | *BBa_I150010 | ||
| + | |||
| + | Programm: | ||
| + | 2min 55/60 | ||
| + | <br/> | ||
| + | <br/> | ||
==<span style="color:orange;">Lysis cassette</span>== | ==<span style="color:orange;">Lysis cassette</span>== | ||
| - | === | + | '''Quickchange PCR''' |
| + | |||
| + | |||
| + | {| style="border-spacing:0;" | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| Name: Theo | ||
| + | |||
| + | |||
| + | |||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| Date: 14.07.2011 | ||
| + | |||
| + | |- | ||
| + | | colspan="2" style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| Continue from Experiment Lysis Cassette | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |- | ||
| + | | colspan="2" style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| Project Name: Correct number of nucleotides between RBS and ATG of temp. sensitive repressor from | ||
| + | |||
| + | Lysis Device | ||
| + | |||
| + | |} | ||
| + | PCR-Mixture for one Reaction: | ||
| + | |||
| + | For a 50 µl reaction use | ||
| + | |||
| + | |||
| + | {| style="border-spacing:0;" | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| 32,5µl | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| H<sub>2</sub>0 | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| Name | ||
| + | |||
| + | |- | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| 10µl | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| 6x HF Phusion Buffer | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| | ||
| + | |||
| + | |- | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| 2.5µl | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| Primer fw | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| P29 (1:10) | ||
| + | |||
| + | |- | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| 2.5µl | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| Primer dw | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| P30 (1:10) | ||
| + | |||
| + | |- | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| 1µl | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| dNTPs | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| | ||
| + | |||
| + | |- | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| 1µl | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| DNA-Template | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| M15a (from S15->diluted to 5ng/µl) | ||
| + | |||
| + | |- | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| 0.5 µl | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| Phusion (add in the end) | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| | ||
| + | |||
| + | |} | ||
| + | What program do you use? | ||
| + | |||
| + | |||
| + | {| style="border-spacing:0;" | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| <center>95°C - 15 sec</center> | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| | ||
| + | |||
| + | |- | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| <center>95°C - 5 min</center> | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| <center>55°C - 15 sec</center> | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| <center>72°C -5 min</center> | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| <center>16°C - ∞</center> | ||
| + | |||
| + | |- | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| <center>72°C - 5’ + 5’’</center> | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| | ||
| + | |||
| + | |- | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| <center>'''20x'''</center> | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| | ||
| + | | style="border:0.0069in solid #00000a;padding-top:0in;padding-bottom:0in;padding-left:0.075in;padding-right:0.075in;"| | ||
| + | |||
| + | |} | ||
| + | |||
| + | <br> | ||
| + | |||
| + | ==<span style="color:grey;">Precipitator</span>== | ||
| + | |||
| + | ===Test-digest=== | ||
| + | |||
| + | '''Investigators: Rüdiger''' | ||
| + | |||
| + | Testdigest of the minipreps, Sophie did (june, 13). | ||
| + | |||
| + | *Enzyme 1: PstI | ||
| + | *Enzyme 2: EcoRI | ||
| + | |||
| + | |||
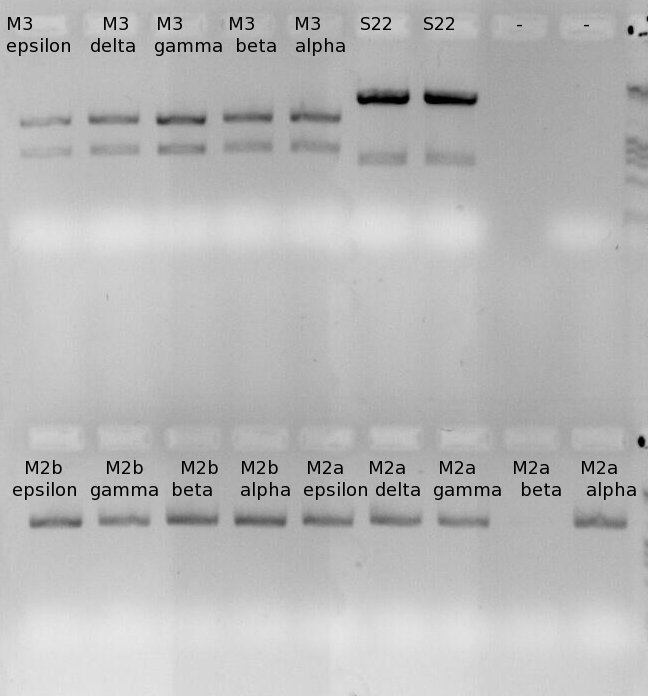
| - | + | [[File:Gimp_Ruediger_13_07.jpg|none|caption]] | |
| + | Image of different samples from miniprep. | ||
| + | {{:Team:Freiburg/Templates/footer}} | ||
Latest revision as of 00:55, 22 September 2011
 "
"
 Contact
Contact