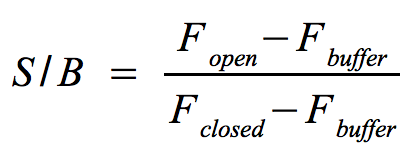Team:Bielefeld-Germany/Protocols/Analytics
From 2011.igem.org


Analytics: This is a list of our used analytical methods.
Bradford Protein Assay
For determination of protein concentrations in protein extracts from bacterial cell lysates or after protein purification steps a Bradford Protein Assay can be applied. For each sample three replicates and at least two dilutions should be measured.
- Prepare differentially concentrated BSA solutions in a range of 0-25 µg/mL for a BSA calibration curve.
- Fill a 96-well plate with 160 µL of each sample.
- Add 40 µL of Bradford Protein Assay Reagent to each well and mix thoroughly by stirring as well as pipetting up and down.
- Incubate for 10 min at room temperature.
- Use a plate reader for the measurement of absorbance at 595 nm.
- Calculate the protein concentration with the help of the BSA calibration curve.
Fluorescence measurements
Measuring of mRFP with Tecan Infinite® M200 platereader
- Take at least 500 µL sample for each measurement (200 µL is needed for one measurement) so you can perform a repeat determination
- Freeze biological samples at -80 °C for storage, keep cell-free at 4 °C in the dark
- To measure the samples thaw at room temperature and fill 200 µL of each sample in one well of a black, flat bottom 96 well microtiter plate (perform at least a repeat determination)
- Measure the fluorescence in a platereader (we used a Tecan Infinite® M200 platereader) with following settings:
- 20 sec orbital shaking (1 mm amplitude with a frequency of 87.6 rpm)
- Measurement mode: Top
- Excitation: 584 nm
- Emission: 620 nm
- Number of reads: 25
- Manual gain: 100
- Integration time: 20 µs
Measuring of mCitrine with Tecan Infinite® M200 platereader
- Take at least 500 µL sample for each measurement (200 µL is needed for one measurement) so you can perform a repeat determination
- Freeze biological samples at -80 °C for storage, keep cell-free at 4 °C in the dark
- To measure the samples thaw at room temperature and fill 200 µL of each sample in one well of a black, flat bottom 96 well microtiter plate (perform at least a repeat determination)
- Measure the fluorescence in a platereader (we used a Tecan Infinite® M200 platereader) with following settings:
- 20 sec orbital shaking (1 mm amplitude with a frequency of 87.6 rpm)
- Measurement mode: Top
- Excitation: 488 nm
- Emission: 529 nm
- Number of reads: 25
- Manual gain: 75
- Integration time: 20 µs
Measuring of mCitrine with Shimadzu RF-5301PC spectrofluorophotometer
- Fill 1 mL sample in a Plastibrand Disposable cuvette 1.5 mL semi-micro PS 12.5 x 12.5 x 45 mm by Brand
- Measure the fluorescence with following settings:
- Exitation: 515 nm
- Emission: 529 nm
- 3 nm exitation filter
- 10 nm emission filter
- sensitivity high or low, depending on fluorescence in sample
Measuring of mCerulean with Tecan Infinite® M200 platereader
- Take at least 500 µL sample for each measurement (200 µL is needed for one measurement) so you can perform a repeat determination
- Freeze biological samples at -80 °C for storage, keep cell-free at 4 °C in the dark
- To measure the samples thaw at room temperature and fill 200 µL of each sample in one well of a black, flat bottom 96 well microtiter plate (perform at least a repeat determination)
- Measure the fluorescence in a platereader (we used a Tecan Infinite® M200 platereader) with following settings:
- 20 sec orbital shaking (1 mm amplitude with a frequency of 87.6 rpm)
- Measurement mode: Top
- Excitation: 433 nm
- Emission: 475 nm
- Number of reads: 25
- Manual gain: 100
- Integration time: 20 µs
Luciferase Measurements
For the luciferase detection we used a Promega Luciferase Assay System, containing a Cell Culture Lysis Reagent, Luciferase Assay Substrate and Luciferase Assay Buffer
For measuring cells:
- Prepare reaction tubes with 10 µL of high salt buffer (1M K2HPO4, 20mM EDTA, pH 7.8)
- Add 90 µL sample, mix and freeze at -80 °C
- For the measurement thaw by placing the tubes in room temperature water
- Add 300 µL of freshly prepared lysis mix (1X Cell Culture Lysis Reagent, 1.25 mg mL-1 lysozyme, 2.5 mg mL-1 BSA, add water for desired volume)
- Mix and incubate the cells for 10 minutes at room temperature
For measuring cell-free samples start at this point:
- Prepare the Luciferase Assay Reagent, by adding 10 mL of Luciferase Assay Buffer to the vial containing the Luciferase Assay Substrate
- Fill each well of a white, flat bottom 96 well microtiter plate with 20 µL of cell lysate
- For the detection of luciferase use a plate reading luminometer with injector for the Luciferase Assay Reagent and following settings (we used a Promega GloMax®-Multi+ Detection System with dual injector):
- Injection volume of Luciferase Assay Reagent: 100 µL
- Delay: 20 secs
- Integration: 3 secs
Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE)
This analytical method can be used for separation and identification of proteins according to their electrophoretic mobility. The mobility is a function of length of the molecular weight. Proteins that have identical charge per unit mass due to binding of SDS results in an equal electrophoretic mobility.
Pouring the polyacrylamide gel
- Make a master mix for the stacking and separating gel without adding ammonium persulfate and TEMED.
- Aliquote 6,5 mL for each separating and 2,5 mL for each stacking gel.
- Add ammonium persulfate and TEMED to each separating gel aliquote and pour the solution quickly into your gel casting form. Leave about 2 centimeters below the bottom of the comb for the stacking gel.
- Layer isopropanol on top of the gel.
- Leave the separating gel at room temperature for >60 minutes to polymerize.
- Remove isopropanol and wait until the surface is dry.
- Add ammonium persulfate and TEMED to each separating gel aliquote and pour the solution quickly into your gel casting form.
- Insert comb without getting bubbles stuck underneath
- Leave the gel at room temperature for >60 minutes to polymerize.
- For storage
- Remove sealing and store the gel wrapped in moistened paper towel at 4°C.
Preparing the sample
- Mix your protein mixture 4:1 with Laemmli-buffer (30 mL protein solution + 10 mL Laemmli-buffer)
- Heat for 5 minutes at 95 °C.
Running the gel
- Remove sealing, put the polymerized gel into gel box and pour SDS running buffer into the negative and positive electrode chamber.
- Remove comp without destroying the gel pocket.
- Pipet the sample into the gel pockets, adjusting the volume according to the amount of protein in your sample. Make sure to include a lane with molecular weight standards (PageRuler Prestained Protein Ladder™ (Fa. Fermentas)) to determinate the molecular weight of your sample.
- Connect the power lead and run the stacking gel with 10 mA until the blue dye front enters the separating gel.
- Raise amperage up to 20 mA for running the separating gel.
- When the distance of the lowest molecular weight standard lane to the gel end is down to 0.5 cm stop the electrophoresis by turning off the power supply.
Polyacrylamide gel staining
Colloidal Coomassie Blue staining
Modified staining protocol from Kang et al., 2002.
- agitate the staining solution at 37 °C over night to form the colloids
- After finishing the SDS-PAGE remove gel from gel casting form and tranfer it in to a box.
- Add 100 mL of the stainig solution to your polyacrylamid gel.
- Incubate the gel in the solution at room temperature until the protein bands got an intensive blue color. Shake the gel continuously during incubation.
- Remove the staining solution
- wash the gel with dH2O
- Incubate the gel in ddH2O (2-6 h) for bleaching the background. Shake the gel continuously during incubation. If necessary replace the colored water with new one.
Fairbanks Coomassie staining
Modiefid steining protocol from Fairbanks et al., (1971).
- After finishing the SDS-PAGE remove gel from gel casting form and tranfer it in to a box with 100 mL of solution A.
- Heat the gel in a microwave for 2 min at 600 W.
- Shake the gel for 5-10 minutes.
- Remove solution A and wash the gel with dH2O.
- Add 100 mL of solution B and heat the gel in a microwave for 2 min at 600 W.
- Shake the gel for 5-10 minutes.
- Remove solution B and wash the gel with dH2O.
- Add 100 mL of solution C and heat the gel in a microwave for 2 min at 600 W.
- Shake the gel for 5-10 minutes.
- Remove solution C and wash the gel with dH2O.
- For bleaching the background add 100 mL of solution D and heat the gel in a microwave for 2 min at 600 W.
- Shake the gel for 5-10 minutes.
- Remove solution D and wash the gel with dH2O. If necessary repeat the last 3 steps with solution D.
Silver staining
- After finishing the SDS-PAGE remove gel from gel casting form and transfer it in to a box.
- Add 50 mL of fixation solution and incubate the gel 1 to 24 h. The formaldehyde (37 %) must be added to the solution short time before.
- Remove the fixation solution and wash the gel 3 times (1-20 min) with 50 mL of 50 % (v/v) ethanol.
- Add 50 mL of thiosulfate solution and incubate the gel exactly for 1 min. If the gel incubates to long the background becomes to dark.
- Remove thiosulfate solution and wash the gel 3 times (20 s) with dH2O.
- Add 50 mL of impregnation solution and icubate the gel for 15 to 20 min. The formaldehyde (37 %) must be added to the solution short time before.
- Remove impregnation solution and wash the gel 2 times (20 s) with dH2O.
- Add 50 mL of fixation solution and incubate the gel until protein bands become visible.
- Remove the developing solution immediately and wash the gel for 10 to 20 s with dH2O.
- Add 50 mL of stop solution and incubate the gel 10 to 20 s.
- Remove the stop solution and wash the gel for 1 min with dH2O.
Tryptic digest of gel lanes for analysis with MALDI-TOF
Note:
- Make sure to work under a fume hood.
- Do not work with protective gloves to prevent contamination of your sample with platicizers.
Reaction tubes have to be cleaned with 60 % (v/v) CH3CN, 0,1 % (v/v) TFA. Afterwards the solution has to be removed completely followed by evaporation of the tubes under a fume hood. Alternatively microtiter plates from Greiner® (REF 650161) can be used without washing.
- Cut out the protein lanes of a Coomassie-stained SDS-PAGE using a clean scalpel. Gel parts are transferred to the washed reaction tubes/microtiter plate. If necessary cut the parts to smaller slices.
- Gel slices should be washed two times. Therefore add 200 µL 30 % (v/v) acetonitrile in 0,1 M ammonium hydrogen carbonate each time and shake lightly for 10 minutes. Remove supernatant and discard to special waste.
- Dry gel slices at least 30 minutes in a Speedvac.
- Rehydrate gel slices in 15 µL Trypsin-solution followed by short centrifugation.
- Gel slices have to be incubated 30 minutes at room temperature, followed by incubation at 37 °C over night.
- Dry gel slices at least 30 minutes in a Speedvac.
- According to the size of the gel slice, add 5 - 20 µL 50 % (v/v) ACN / 0,1 % (v/v) TFA.
- Samples can be used for MALDI measurement or stored at -20 °C.
Trypsin-solution: 1 µL Trypsin + 14 µL 10 mM NH4HCO3
- Therefore solubilize lyophilized Trypsin in 200 µL of provided buffer and incubate for 15 minutes at 30 °C for activation. For further use it can be stored at -20 °C.
Preparation and Spotting for analysis of peptides on Bruker AnchorChips
- Spot 0,5 - 1 µL sample aliquot
- Add 1 µL HCCA matrix solution to the spotted sample aliquots. Pipet up and down approximately five times to obtain a sufficient mixing. Be careful not to contact the AnchorChip.
Note: Most of the sample solvent needs to be gone in order to achieve a sufficiently low water content. When the matrix solution is added to the previously spotted sample aliquot at a too high water content in the mixture, it will result in undesired crystallization of the matrix outside the anchor spot area.
- Dry the prepared spots at room temperature
- Spot external calibrants on the adjacent calibrant spot positions. Use the calibrant stock solution (Bruker’s “Peptide Calibration Standard II”, Part number #222570), add 125 µL of 0.1% TFA (v/v) in 30% ACN to the vial. Vortex and sonicate the vial.
- Mix the calibrant stock solution in a 1:200 ratio with HCCA matrix and deposit 1 µL of the mixture onto the calibrant spots.
Bisphenol A analysis
Extraction with ethylacetate
- mix 100 µL culture supernatant with 100 µL internal standard ( bisphenol F, 100 µg L-1)
- add 200 µL ethylacetate (HPLC grade) for extraction
- vortex (30 s)
- centrifuge for phase separation (5 min, 5000 g)
- take a bit from upper phase and put it in a clean reaction tube
- SpeedVac at 40 °C to remove ethlyacetate
- solve remaining BPA in water (HPLC grade), vortex (30 s)
- solubility of BPA in water only 300 mg L-1
- for LC-MS analysis of BPA, 300 mg BPA L-1 is definitely enough
- if you want to detect or expect higher concentrations of BPA, solve it in an acetonitrile-water-mix
HPLC method
- C18 reverse phase column
- Isocratic method: 45 % Acetonitrile
- Flow = 0.6 mL min-1
- UV-detection at 227 nm
- Internal standard: 100 mg L-1 Bisphenol F (BPF)
- Column:
- Eurospher II 100-5 C18p by Knauer
- Dimensions: 150 x 4.6 mm with precolumn
- Particle size: 5 µm
- Pore size: 100 Å
- Material: silica gel
- Software:
- Clarity (Version 3.0.5.505) by Data Apex
- Autosampler:
- Midas by Spark Holland
- Tray cooling: 10 °C
- Pump:
- L-6200A Intelligent Pump by Hitachi
- UV-Detector:
- Series 1050 by Hewlett Packard
LC-ESI-qTOF-MS(-MS)
HPLC method
- Column: C18 reverse phase column (Knauer Blue Orchid)
- dimension: 50 x 2 mm
- Pore size: 175 Å
- Particle size: 1.8 µm
- Flow: 0.4 mL min-1
- Column temperature: 30 °C
- Gradient:
- 0 - 1.05 min: 45 % acetonitrile
- 2.55 min: 95 % acetonitrile
- 6.00 min: 95 % acetonitrile
- 6.15 min: 45 % acetonitrile
- 12.00 min: 45 % acetonitrile
- VWR Hitachi LaChrom ULTRA HPLC equipment
- Software: HyStar 3.2, HyStarPP, mircrOTOF Control
Ionization method
- Using Bruker Daltonics micrOTOFQ
- ESI in negative mode
- Mass range: 50 - 1500 m/z
- End plate offset: - 500 V, 107 nA
- Capillary: 2500 V, 4 nA
- Nebulizer: 3 bar
- Dry gas: 8 L min-1
- Quadrupole
- Ion energy: 5 eV
- Low mass: 100 m/z
- Collision energy: 10 eV
- Collision RF: 150 Vpp
- Transfer time: 70 µs
- Pre puls storage: 7 µs
MS-MS
- Isolated mass: 243.1 +/- 0.1
- Collision energy: 30 eV
NAD+ detection
Characterization of molecular beacons
Molecular beacons are nucleic acid probes that fluoresce upon hybridization to a complementary target. To check whether they are suitable for a NAD+ bioassay involving NAD+-dependent DNA ligase from Escherichia coli (LigA) they can be characterized by thermal profile analysis and determination of the signal-to-background ratio. Detailed background information, tools for the correct design of molecular beacons, a list of synthesis companies as well as protocols for the synthesis and characterization of molecular beacons can be found here. The used nucleic acids were designed as follows:
| Name | Sequence | Label |
|---|---|---|
| Molecular beacon | 5'-CCT CTC CGT GTC TTG TAC TTC CCG TCA GAG AGG-3' | 5'-FAM, 3'-Dabcyl |
| Target | 5'-GAC GGG AAG TAC AAG ACA C-3' | |
| Split target | 5'-TAC AAG ACA C-3' | 5'-phosphate |
| 5'-GAC GGG AAG-3' |
The molecular beacon was purchased from Eurogentec and labeled with 6-FAM (fluorophor) and Dabcyl (appropriate quencher for 6-FAM). Preferentially, use a complementary oligonucleotide (target) as well as two enshortened oligonucleotides (split target) for characterizing the molecular beacon in its closed and open state. In this way you can verify simultaneously the ineffectiveness of the split target to reach the molecular beacons open state.
- Optimal wavelengths:
- Determine the emission spectrum of your molecular beacon by initially using a literature value for the exctintion wavelength of the labeled fluorophore. For this add 10 µL of 1 µM molecular beacon in 200 µL NAD+ bioassay buffer, mix thoroughly and wait 2 min until measuring the fluorescence with a spectrofluorometer at an defined temperature (optimally 37 °C) .
- Add 10 µL of 2 µM either target or split target and shake at least 6 min until measuring the fluorescence again (when equilibrium is reached).
- Repeat the procedure to determine the extinction spectrum using the self-calculated optimal emission wavelength for your fluorophore/quencher combination. For further fluorescence measurements use the extinction wavelength for which the difference between the molecular beacons closed state and the open state signals is maximal.
- Thermal profile analysis:
- Prepare a 30 µL reaction mix composed of NAD+ bioassay buffer and 1 µM molecular beacon. In a separate sample add 2 µM either target or split target.
- Use a spectrofluorometric thermal cylcer to dertemine the fluorescence as a function of temperature. Start at 80 °C and decrease the temperature stepwise in 1 °C intervals holding each temperature for 1 min and measuring the fluorescence at the end of each step.
- Signal-to-background ratio (S/B):
- The approach is quite similar to the one that determines the optimal wavelengths which should be used to monitor the fluorescence of the NAD+ bioassay buffer (Fbuffer) and the added molecular beacon (Fclosed) as well as the target (Fopen) or split target. Monitor the fluorescence at the optimal wavelengths until the equilibrium is reached each time before you add a new component.
- The signal-to-background ratio can be calculated as shown on the right. Finally, compare the signal-to-background ratios after adding the target and split target to check whether the split target has an effect on the melting of the molecular beacons hairpin structure.
- Imaging:
- Prepare a 200 µL reaction mix composed of NAD+ bioassay buffer and 500 nM molecular beacon in a PCR tube. In a separate sample add 600 µM either target or split target. Excitate the samples by a UV transilluminator and take images preferably with a camera that can detect different colours.
Purification of DNA ligase
The purification of the overexpressed NAD+-dependent DNA ligase gene (<partinfo>K525710</partinfo>) in E. coli was performed under native conditions and Ni-NTA columns were used utilizing the recombinant protein`s C-terminal 6xHis-tag.
- Cultivation
- Prepare an overnight culture of E. coli KRX carrying the plasmid with DNA ligase (<partinfo>K525710</partinfo>) in 30 mL LB medium containing 20 µg mL-1 chloramphenicol at 37 °C.
- Dilute the overnight culture in 100 ml autoinduction medium (20 µg mL-1 chloramphenicol added) to an OD600 = 0.1 and harvest the cells after 4 h growth at 37 °C.
- Use the centrifuged cells immediately for protein purification or store them at -20 °C.
- Small-scale purification
- Resuspend a pellet derived from 5 mL cell culture volume in 630 µL NPI-10.
- Add 70 µl lysozyme (10 mg/mL) as well as 25 Units benzonase nuclease and mix thoroughly.
- Incubate on ice for 30 min.
- Centrifuge the lysate for 30 min at 12000 g (4 °C). Collect the supernatant (cleared lysate).
- Prepare a Ni-NTA spin column (QIAGEN) by equilibration with 600 µl NPI-10 and centrifugation for 2 min at 890 g (4 °C).
- Load up to 600 µl cleared lysate onto the Ni-NTA spin column and centrifuge for 5 min at 270 g (4 °C). Collect the flow-through.
- Wash the Ni-NTA spin column with 600 µL NPI-20 and centrifuge for 2 min at 890 g (4 °C). Repeat this step and collect flow-through each time.
- Elute the protein with 300 µL NPI-500 and centrifuge for 2 min at 890 g (4 °C). Repeat this step and collect flow-through each time.
- For centrifugal ultrafiltration use Vivaspin 500 concentrators (Sartorius) with a 5000 molecular weight cut-off PES membrane. Repeat the procedure three times by adding DNA ligase buffer (4 °C) for buffer exchange (remove of imidazole).
- Check each purification step and especially the purity of the protein in the final condition by SDS-PAGE analysis.
- Determine the protein concentration with a Bradford Protein Assay.
- For long-time storage keep the protein at -20 °C.
- If the binding conditions are not effective enough you can reduce the imidazole concentration in NPI-10 to 1-5 mM. If the eluate contains unspecifically bound proteins maybe a washing step with NPI-10 containing 50 mM or 100 mM imidazole could help to get higher purity.
- Large-scale purification
- For production of DNA ligase with high yield see chapter His-tag affinity chromatography.
Deadenylation
The majority of purified DNA ligase from E. coli KRX is usually in the adenylated form. But only the apoenzyme is useful for the NAD+ detection so that the AMP moiety has to be removed.
- Mix the purified DNA ligase with deadenylation buffer and incubate for 20 min at 37 °C.
- For centrifugal ultrafiltration use Vivaspin 500 (Sartorius) concentrators with a 5000 molecular weight cut-off PES membrane. Repeat the procedure several times by adding DNA ligase buffer (remove of NMN and formed NAD+).
- Check the deadenylation process by SDS-PAGE analysis with high resolution. If the deadenylation was successful there should not be a double band indicating both forms of DNA ligase.
- Determine the protein concentration with a Bradford Protein Assay.
- For long-time storage keep the protein at -20 °C.
NAD+ bioassay
- Prepare a master mix composed of 250 nM molecular beacon and 250 nM split target (notice that the 5'-end at the nick site has to be phosphorylated) in NAD+ bioassay buffer.
- Incubate for 20 min at 37 °C.
- Add 500 ng purified and deadenylated DNA ligase (final concentration 5 ng/µL).
- Monitor the fluorescence with a spectrofluorometer at 37 °C and wait until it reaches equilibrium.
- Add 1 µl NAD+ in different final concentrations (e.g. 0, 5, 10, 20, 30, 50, 80, 100, 200, 300, 500, 1000, 10000 nM) filling the reaction mix up to 100 µL, stirr shortly and monitor the fluorescence at 37 °C again.
- Determine the initial enhancement rate of fluorescence intensity for each NAD+ concentration (in a range of 200 s after NAD+ addition).
- For the calibration curve plot the initial velocity versus the NAD+ concentration.
Imaging
- It is a similar procedure to the one mentioned above except that you prepare 500 nM molecular beacon and 600 nM target or split target dissolved in bioassay buffer in a PCR tube.
- After incubation at 37 °C add 1000 ng DNA ligase and possibly NAD+ in different concentrations filling the reaction mix up to 200 µL.
- Visualize the fluorescence in a laser scanner or UV transilluminator and document the results.
Selectivity test
- Follow the instructions for the NAD+ bioassay but use different analytes like NADH, NADP+, NADPH, 3-APAD, ATP, ADP or AMP in a final concentration of 100 nM.
Coupled enzyme reaction
- To check whether the NAD+ bioassay is suitable for the analysis of NADH-dependent enzymatic reactions it was coupled with the NADH-dependent conversion of pyruvate to L-lactic acid by lactic acid dehydrogenase (LDH) from E. coli.
- Prepare a master mix composed of 250 nM molecular beacon and 250 nM split target (notice that the 5'-end at the nick site has to be phosphorylated) in NAD+ bioassay buffer (50 mM Tris-HCl pH 8.0, 10 mM MgCl2, 2.5 mM CaCl2, 5 mM DTT, 0.05 % BSA).
- Incubate for 20 min at 37 °C.
- Meanwhile, prepare a separate reaction mix composed of LDH reaction buffer (20 mM Tris-HCl pH 7.5, 50 mM NaCl), 100 µM NADH and 50 ng/µL LDH.
- Incubate for 5 min at 37 °C.
- Add 650 ng purified and deadenylated LigA to the molecular beacon reaction mix (final concentration 6.5 ng/µL).
- Monitor the fluorescence with a spectrofluorometer at 37 °C and wait until it reaches equilibrium.
- Add 1 µL pyruvate in different final concentrations (e.g. 0, 1, 2.5, 5, 7.5, 10 µM) to the LDH reaction mix.
- Incubate for 2 min at 37 °C.
- Add 1 µL LDH reaction mix to the equilibrated NAD+ bioassay solution filling the reaction mix up to 100 µL, stirr shortly and monitor the fluorescence at 37 °C again.
- Determine the initial enhancement rate of fluorescence intensity for each pyruvate concentration (in a range of 100 s after LDH reaction mix addition).
- For the calibration curve plot the initial velocity versus the pyruvate concentration.
 "
"